 In June of 2016, the Council of the European Union invited the European Commission to prepare “an evidence based analysis” of the impact of the incentives on innovation of medicinal products. The request noted that, particular attention should be given to the purpose of Supplementary Protection Certificates (SPCs) and the market exclusivity for orphan medicinal products. As Brussels continues the review of these incentives, it is worth reflecting on several recent studies that illuminate the impact of SPCs and their importance in fostering innovation.
In June of 2016, the Council of the European Union invited the European Commission to prepare “an evidence based analysis” of the impact of the incentives on innovation of medicinal products. The request noted that, particular attention should be given to the purpose of Supplementary Protection Certificates (SPCs) and the market exclusivity for orphan medicinal products. As Brussels continues the review of these incentives, it is worth reflecting on several recent studies that illuminate the impact of SPCs and their importance in fostering innovation.
In 1992, the European Economic Community (EEA) enacted Supplementary Protection Certificates under Council Regulation No. 1768/92, the “SPC Regulation”. The purpose of the regulation was to harmonize the scope of SPC protection in the EU with the ultimate goals of promoting pharmaceutical research in the European Union and creating a simple, transparent application system. In the decades since implementation, several trends emerge. These are described in recent papers by Mejer (May 2017) and Kyle (January 2017). The trends demonstrate that there is a need for SPC Regulation in order to ensure that biopharmaceutical innovation continues to be incentivized and rewarded. This growing need for SPC is echoed in the increased use of the provision, across products and Member States. Specifically, the study finds:
- Over the period between 1990 and 2015, development times for pharmaceuticals have increased by more than two years on average, while the same period also corresponds to a drop of 1.4 years in the lag between the first global launch and the first EU launch. Accordingly, the remaining patent term is almost a year less for products introduced since 2010, relative to those introduced in the early 1990s.
- On average, innovators receive 12.46 years of protection from basic patents and SPCs.
- The use of SPC protection has significantly increased since 1993. (Figure 1, below)
- Currently, SPC protection for a single medicinal product is filed in 20 EU Member States on average. (Figure 2, below)
- Today, a higher share of products, now 86%, is covered by SPCs.
All of these trends point to the increasing importance of SPC and the vital role they play in the European Union’s industrial competitiveness.
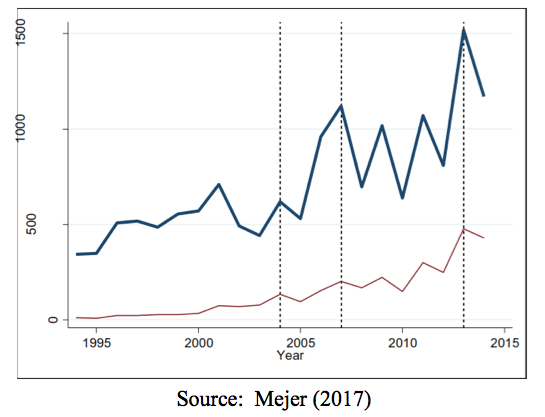
Figure 1: Number of SPC Applications in the EU, by first marketing authorization year. Notes: The blue bold line presents the number of SPC applications in the EU28 and the thin red line the number of SPC applications in all 13 Member States that joined in 2004 and after. A vertical dashed line indicates subsequent enlargements in 2007 and 2013.
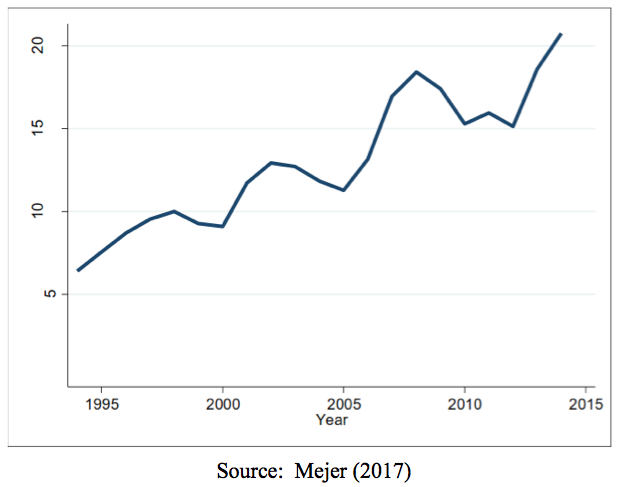
Figure 2: Average number of countries where SPC protection is applied for the same product. Note: The line presents average number of EU Member States where SPC related to the same product was applied for.
For the biopharmaceutical industry, drug development is an expensive, time-consuming, risky process. It is characterized by tremendous fixed costs, especially in the case of therapies requiring lengthy, large-scale clinical trials. Drug development requires substantial resources, in terms of time, talent and financial capital, as described by Kyle (2017):
“In a competitive market, the incentives to invest in R&D are minimal: the firm that incurs the development costs creates a public good – namely the identification of a safe and effective drug – but competition from imitators drives prices down to marginal costs, leaving the investor unable to recoup the development costs. Patent protection is one solution to this incentive problem. With entry restricted for the duration of the patent, the investing firm has an opportunity to sell at prices that exceed production costs.”
Upon reaching the market, drugs have less than the 20 year term of protection provided by a patent. Moreover, this deficit is greater for therapies for diseases for which drug development requires longer periods of time. As a consequence, innovators may reduce investment in researching those diseases and conditions since they have fewer years of effective patent protection remaining during which to recoup their R&D costs (Budish, Roin & Williams, 2015; Lietzan, 2017). Enter the SPCs. SPCs are able to partly offset the reduction in R&D incentives that stem from longer development times. In essence, SPCs endeavor to correct a distortion in research incentives generated by differences in development times across products. Moreover, SPCs come at low cost – governments spend very little to implement and grant them.
The emerging trends over the past 25 years suggest SPCs are increasingly important to encouraging biopharmaceutical innovation and investment in the EU. As such, reducing the scope or duration of IP incentives risks jeopardizing the EU’s competitiveness, job creation and growth.
First, the European Union suffers from an investment deficit relative to other industrialized nations. A recent report by the European Commission emphasizes this impact, “the EU needs to put in place better incentives and conditions for businesses to innovate” in important areas such as market regulations, intellectual property rights protection, barriers to entrepreneurship, and ease of doing business. Given this, encouraging investment is essential to future growth. Weakening the IP incentives embedded in SPCs would be a step in the wrong direction.
Second, these protections and the investments it stimulates have direct economic consequences for the European Union. The benefits are significant, and wide-reaching. Recent studies by the American Chamber of Commerce to the European Union and by the Pugatch Concilium demonstrate that the direct impacts include:
- 38% of the 82 million jobs in Europe are directly and indirectly employed in IP-intensive industries.
- 42% of the EU’s GDP is generated by IP-intensive industries, which account for approximately 90% of EU’s trade with other countries.
- The biopharmaceutical industry supports over two million jobs in the EU.
- In the first decade of implementation, the Orphan Medicinal Products Regulation resulted in a 30% increase in new biotech companies across Europe.
- Between 2001 and 2016, the number of orphan drug designations in the EU, US and Japan has increased from 150 to 557.
- As of December 2016, EU designations for rare diseases have resulted in authorized medicinal products for 101 conditions.
- Between 1982 and 2003, only two percent of NCEs (new chemical entities) were developed outside the EU, US and Japan – the jurisdictions with the strongest and clearest IP incentives and protections in place. Essentially, these three jurisdictions are responsible for the vast majority of biopharmaceutical innovation.
Two things are clear: biopharmaceutical innovation undeniably relies on the availability of strong, effective IP incentives and the competitiveness of the EU biopharmaceutical sector is vital to the economic health of the Member States, growth, and job creation. Instead of weakening the incentives for innovation in this critical industry, Brussels should be exploring mechanisms for expanding, enhancing and magnifying these inducements. It seems that the writing is on the wall: given the potential benefits through the economic impact, the public health consequences, and the industry effects – for patients, workers, firms, and national economies – policymakers should embrace stronger incentive and increased innovation.

![[IPWatchdog Logo]](https://ipwatchdog.com/wp-content/themes/IPWatchdog%20-%202023/assets/images/temp/logo-small@2x.png)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Patent-Litigation-Masters-2024-sidebar-early-bird-ends-Apr-21-last-chance-700x500-1.jpg)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/WEBINAR-336-x-280-px.png)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/2021-Patent-Practice-on-Demand-recorded-Feb-2021-336-x-280.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/Ad-4-The-Invent-Patent-System™.png)







Join the Discussion
No comments yet.