“Out of the 230 Orange Book patents challenged in IPR proceedings, 90.4% (208) of these patents were also challenged in Hatch-Waxman litigation…”
 More than three decades ago, Congress enacted the Hatch-Waxman Act, which provided a streamlined pathway to challenge validity of Orange Book patents in federal district courts. The Hatch-Waxman Act intended to strike a balance between encouraging pioneer drug innovation by offering pioneers patent term restoration and data exclusivity and promoting market entry of affordable generic versions of pioneer drugs by giving generic manufacturers a lucrative 180-day marketing exclusivity period if they are the first filer to initiate a Paragraph IV Abbreviated New Drug Application (ANDA) challenge against an Orange Book drug patent.
More than three decades ago, Congress enacted the Hatch-Waxman Act, which provided a streamlined pathway to challenge validity of Orange Book patents in federal district courts. The Hatch-Waxman Act intended to strike a balance between encouraging pioneer drug innovation by offering pioneers patent term restoration and data exclusivity and promoting market entry of affordable generic versions of pioneer drugs by giving generic manufacturers a lucrative 180-day marketing exclusivity period if they are the first filer to initiate a Paragraph IV Abbreviated New Drug Application (ANDA) challenge against an Orange Book drug patent.
In 2012, the America Invents Act (AIA) introduced Inter Partes Review (IPR) proceedings in 2012 as a new avenue for challenging patents before the Patent Trial and Appeal Board (PTAB). IPRs were intended to improve patent quality by offering an efficient lower cost pathway to challenge weaker patents. Today, over 7,600 IPR petitions have been filed with the PTAB, making IPRs a very popular pathway for challenging patent validity. Over 400 of these IPRs filed challenged patents listed in the Food and Drug Administration’s (FDA’s) “Approved Drug Products with Therapeutic Equivalence Evaluations” publication, commonly known as the Orange Book. Recently in Oil States and SAS, the Supreme Court has affirmed the constitutionality of IPR proceedings and held that the PTAB must institute trials on every claim the petitioner has challenged, as the PTAB is not authorized to carry on their practice of so-called “partial institution” by instituting trials in only some of the challenges raised in the petition. This expansion of the scope of institution in IPRs is likely to encourage even more IPR challenges against Orange Book patents in the future.
Some scholars fear that IPRs would disrupt the delicate balance the Hatch-Waxman Act intended to strike and deter pioneer drug innovation by offering a faster and cheaper way to invalidate Orange Book patents, which requires a lower evidentiary burden of proof and a broader claim construction standard, making it easier, in theory, to obtain patent invalidation as compared to Hatch-Waxman district court litigation. Out of this growing concern, Senator Hatch has even proposed an amendment that would require generic manufacturers to choose between litigating patent validity in Hatch-Waxman district court litigation or via AIA challenges such as IPRs. To help evaluate the relationship between IPRs and Hatch-Waxman litigation, I have conducted an empirical study on IPR outcomes of all 412 IPRs filed against 230 Orange Book patents between 03/16/2012 and 03/16/2018 (my study database is publicly available). This empirical study assesses whether the IPR pathway is disrupting the delicate Hatch-Waxman balance by allowing patent challenges to occur in ways not possible in district court, such as during the data exclusivity periods before an ANDA can be filed. The study finds that generic manufacturers are not targeting Orange Book patents in IPRs during their drug exclusivity period since so far generic petitioners have filed only 2 IPRs against Orange Book patents during their New Chemical Entity exclusivity period.
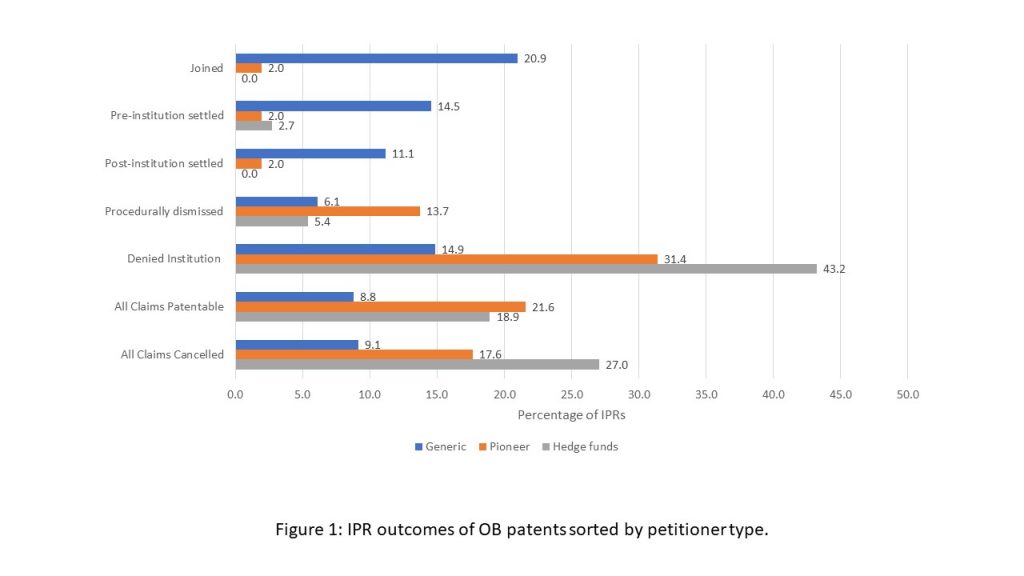
FIGURE 1: IPR outcomes of OB patents sorted by petitioner type.
Generic manufacturers were able to obtain more patent invalidation outcomes but settled less with pioneer drug manufacturers in IPR proceedings than Hatch-Waxman litigation. Figure 1 shows that in IPR proceedings, generic manufacturers obtained all claims cancelled decisions in 9.1% of the IPR petitions filed, whereas in Hatch-Waxman litigation, generic manufacturers won only 4.1% of their cases. However, at the same time, IPRs filed against Orange Book patents resulted in more favorable outcomes for patent owners (23.7% of IPRs result in favorable final written decisions or are denied institution) than Hatch-Waxman litigation (15%). Generic manufacturers settled with pioneer patent owners in 25.6% of the IPRs filed, while settlement was observed in 45% of the Hatch-Waxman cases.
Out of the 230 Orange Book patents challenged in IPR proceedings, 90.4% (208) of these patents were also challenged in Hatch-Waxman litigation perhaps due to the lucrative 180-day exclusivity incentive available to the first generic manufacturer to file a paragraph IV challenge when the Orange Book drug patent is successfully invalidated in a subsequent district court proceeding. Therefore, the IPR process has provided generic manufacturers a dual track option for challenging Orange Book patents by initiating Hatch-Waxman litigation and also pursuing IPRs. Overall, because the rate of settlement in IPRs is much lower than in Hatch-Waxman litigation, both generic manufacturers and patent owners obtain more favorable final decisions in IPRs as compared to their Hatch-Waxman litigation outcomes. Thus, this study suggests that IPRs do not appear to deter generic manufacturers from Hatch-Waxman litigation or disrupt the fine balance it strikes between encouraging medical innovation and providing access to affordable drugs.
Scholars have also voiced their concern about hedge funds, which do not have standing to challenge Orange Book patents in federal district courts, using the IPR process to invalidate patents. Hedge funds could potentially file IPRs against Orange Book patents while short-selling securities in the patent owners’ companies. However, Figure 1 shows that only 9% (37) of IPRs filed against Orange Book patents were filed by hedge funds, 34 of which were filed by Kyle Bass and Erich Spangenberg hedge fund, Coalition for Affordable Drugs. About 43.2% of IPRs filed by hedge funds were denied institution on the merits as compared to only 14.9% of those filed by generic petitioners. But at the same time, both hedge funds and generic petitioners obtain similar percentages of favorable final written decisions from the PTAB as unfavorable ones. Unlike generic petitioners, who settle in 25.6% IPRs, hedge funds settled with patent owners in only 2.7% (1 out of 37) IPRs. One explanation for this striking difference in settlement rates could be that hedge funds are primarily motivated to invalidate patents so that they can profit from short sell securities in patent owners’ companies, which makes them less likely to settle with patent owners. Overall, the data seems to suggest that hedge funds are probably not filing frivolous IPRs challenges against Orange Book patents on a large scale.
In conclusion, IPR proceedings do not appear to be deterring generic manufacturers from Hatch-Waxman litigation or disrupting the delicate balance sought by the Hatch-Waxman Act between encouraging medical innovation and providing access to affordable drugs. Instead, the IPR process has provided generic manufacturers a dual track option for challenging Orange Book patents by initiating Hatch-Waxman litigation in district courts and also pursuing patent invalidity in IPRs before the PTAB, which has reduced rate of settlements resulting in more patents being upheld and invalidated.
For a more complete account of IPR Outcomes of Orange Book Patents and its Effect on Hatch-Waxman Litigation, please read a draft of my article Maintaining the Balance: An Empirical Study on Inter Partes Review Outcomes of Orange Book Patents and its Effect on Hatch-Waxman Litigation. Because the article is still in draft format, I welcome any comments or suggestions via e-mail. I have also made my empirical study database publicly available for further research purposes.

![[IPWatchdog Logo]](https://ipwatchdog.com/wp-content/themes/IPWatchdog%20-%202023/assets/images/temp/logo-small@2x.png)

![[[Advertisement]]](https://ipwatchdog.com/wp-content/uploads/2024/04/Patent-Litigation-2024-banner-938x313-1.jpeg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/UnitedLex-May-2-2024-sidebar-700x500-1.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Artificial-Intelligence-2024-REPLAY-sidebar-700x500-corrected.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Patent-Litigation-Masters-2024-sidebar-700x500-1.jpg)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/WEBINAR-336-x-280-px.png)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/2021-Patent-Practice-on-Demand-recorded-Feb-2021-336-x-280.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/Ad-4-The-Invent-Patent-System™.png)






Join the Discussion
3 comments so far.
Xtian
October 5, 2018 09:08 am@Mahaseth,
Thanks for your response. Here’s my experience between IPRs and HW litigation involving generics and branded pharma:
Assume 5 year NCE and at least one patent that expires years after NCE exclusivity.
Drug approved year 1 – patents listed in Orange Book.
year 4 – ANDAs are filed by generics, Brand sues generics, litigation starts;
year 5 – (HW litigation is in discovery/expert deposition stage); generics file IPR on OB patents (wihtin their 1 year time prior after litigation)
year 6 – HW litigation continues; maybe though a markman hearing; meanwhile IPR decision to institute either happens or does not happen.
If IPR instituted – request a stay in HW litigation pending IPR outcome.
If IPR not instituted – Brand has upper hand in HW litigation; brand offers settlement rather than fighting it out to close the litigation.
There is no balance….
Tulip Mahaseth
October 4, 2018 11:14 amHi xtian,
Thanks for your comments. The study’s conclusion that IPRs do not appear to deter generic manufacturers from Hatch-Waxman litigation or disrupt the fine balance it intended to strike is based on the following data:
1. Out of the 230 Orange Book patents challenged in IPR proceedings, 90.4% (208) of these patents were also challenged in Hatch-Waxman litigation. So generics are not exclusively challenging Orange Book patents through the IPR pathway, making the ANDA process obsolete.
2. Despite this dual track system to challenge patents, the data shows that IPRs filed against Orange Book patents resulted in more favorable outcomes for patent owners (23.7% of IPRs result in favorable final written decisions or are denied institution) than Hatch-Waxman litigation (15%). But at the same time, as you noted, generic manufacturers were also able to obtain more patent invalidation outcomes but settled less with pioneer drug manufacturers in IPR proceedings than Hatch-Waxman litigation. In IPR proceedings, generic manufacturers obtained all claims cancelled decisions in 9.1% of the IPR petitions filed, whereas in Hatch-Waxman litigation, generic manufacturers won only 4.1% of their cases. Generic manufacturers settled with pioneer patent owners in 25.6% of the IPRs filed, while settlement was observed in 45% of the Hatch-Waxman cases.
So overall, IPRs reduce the rate of settlement and lead to more patents being upheld and invalidated, thereby mataining the fine balance the Hatch-Waxman Act intended strike between drug innovation and affordability.
3. There has been some concern that IPRs would allow generic manufacturers to challenge and invalidate Orange Book patents during their NCE period when ANDA litigation could not be commenced. But as you noted, out of the 412 IPRs against Orange Book patents, only 2 were filed during the drugs’ NCE period. So IPRs are not disrupting the ANDA Process as Orange Book patents are not being challenged in IPRs before ANDA litigation could be commenced in district court. It is possible that the benefits of obtaining 180 day exclusivity is likely keeping the generic manufacturers from challenging Orange Book patents during the NCE period.
xtian
October 4, 2018 09:36 am“this study suggests that IPRs do not appear to deter generic manufacturers from Hatch-Waxman litigation or disrupt the fine balance it strikes between encouraging medical innovation and providing access to affordable drugs.”
So adding a new pathway to challenge patents didn’t effect the balance? The author admits that generics don’t go after the OB patents right away: “The study finds that generic manufacturers are not targeting Orange Book patents in IPRs during their drug exclusivity period since so far generic petitioners have filed only 2 IPRs against Orange Book patents during their New Chemical Entity exclusivity period.”
Any why would they? If a generic successfully challenges a patent in an IPR during the data exclusivity period, then the 180 award to the generic for challenging the OB patent in a HW case go away. Why would a generic destroy their on potential for a 180 day duopoly?
As the author admits, generic companies wait until they they are sued by the Brand in a HW case (year 4 of 5 in NCE cases), then files the IPR in the hopes that the IPR is an easier forum to take down the patent. To wit: “Generic manufacturers were able to obtain more patent invalidation outcomes but settled less with pioneer drug manufacturers in IPR proceedings than Hatch-Waxman litigation.”
So I have a hard time squaring the author’s conclusion….