“[I]t appears that the pendulum is swinging back in favor of Orange Book patent owners. The number of petitions challenging these patents has slowed since the high rates seen in the 2015 and 2016 fiscal years. The rate of institution denials of Orange Book patents has risen considerably…”
 Since its inception, the Patent Trial and Appeal Board (PTAB) has been a frequent venue for patent challenges in the pharmaceutical and biotechnology industries. By the end of the U.S. Patent and Trademark Office’s (USPTO’s) 2018 fiscal year, patents in those fields were targeted in nearly 10% of all petitions for inter partes review (IPR), totaling approximately 900 individual petitions. Of these 900 petitions, roughly 5% challenged patents listed in the FDA’s Orange Book for approved drug products. The remaining petitions challenged biologic drugs (1.3%) and other biologic-, biotechnology-, or pharmaceutical-related patents (3.5%). Many of these petitions have ultimately resulted in the cancellation of all challenged claims, including those of a significant number of Orange Book patents. Based on the PTAB’s initial high rate of claim cancellation in pharma and other areas, critics of the PTAB were quick to deem it a patent “death squad.” Does the PTAB still deserve the “death squad” label when it comes to Orange Book patents? In this article, we examine the rates of challenge, institution, and final written decision outcomes for patents listed in the Orange Book, from the PTAB’s inception through the end of its 2018 fiscal year.
Since its inception, the Patent Trial and Appeal Board (PTAB) has been a frequent venue for patent challenges in the pharmaceutical and biotechnology industries. By the end of the U.S. Patent and Trademark Office’s (USPTO’s) 2018 fiscal year, patents in those fields were targeted in nearly 10% of all petitions for inter partes review (IPR), totaling approximately 900 individual petitions. Of these 900 petitions, roughly 5% challenged patents listed in the FDA’s Orange Book for approved drug products. The remaining petitions challenged biologic drugs (1.3%) and other biologic-, biotechnology-, or pharmaceutical-related patents (3.5%). Many of these petitions have ultimately resulted in the cancellation of all challenged claims, including those of a significant number of Orange Book patents. Based on the PTAB’s initial high rate of claim cancellation in pharma and other areas, critics of the PTAB were quick to deem it a patent “death squad.” Does the PTAB still deserve the “death squad” label when it comes to Orange Book patents? In this article, we examine the rates of challenge, institution, and final written decision outcomes for patents listed in the Orange Book, from the PTAB’s inception through the end of its 2018 fiscal year.
Analysis
For our analysis, we subdivided challenged patents into categories based on how they were listed in the Orange Book. About 15% of challenged patents were compound patents, 38% were formulation patents, and 48% were method of treatment patents. The rates at which these categories of patents have been challenged cannot be explained by their Orange Book listing rates alone. For comparison, from 2014 through the end of the 2018 fiscal year, Orange Book listed patents comprised 21% compound patents, 44% formulation patents, and 35% method of treatment patents—i.e., in PTAB challenges, compound and formulation patents were underrepresented, while method of treatment patents were overrepresented. Patent challengers may perceive method of treatment claims as more susceptible to anticipation and obviousness challenges compared to compound and formulation patents, making them more attractive targets for challenge. This theory is possibly borne out by the relatively higher institution rates for method of treatment claims, as discussed further below. Method of treatment patents may also be targeted over compound patents because they are often filed later in the drug development process and therefore expire later.
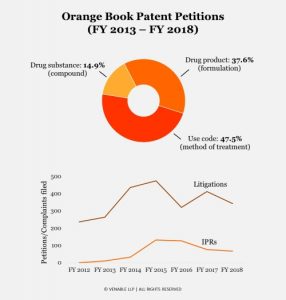 Post-grant challenges to Orange Book patents got off to a relatively slow start. Only 10 petitions on Orange Book patents were filed in 2013 and 32 in 2014. Then, there was a surge of petitions over the 2015 fiscal year (132 petitions), and this high level was maintained in 2016 (127 petitions). Petitions declined in the 2017 fiscal year (76 petitions) and again dipped slightly in 2018 (67 petitions).
Post-grant challenges to Orange Book patents got off to a relatively slow start. Only 10 petitions on Orange Book patents were filed in 2013 and 32 in 2014. Then, there was a surge of petitions over the 2015 fiscal year (132 petitions), and this high level was maintained in 2016 (127 petitions). Petitions declined in the 2017 fiscal year (76 petitions) and again dipped slightly in 2018 (67 petitions).
The surge in petitions in 2015 and 2016 was largely attributable to the significant number of challenges (37 petitions) filed by hedge funds during that period. The downward trend observed in 2017 and 2018 could be explained by financial pressures on the generic pharmaceutical industry or reactions to trends in institution or final written decision rates, which are discussed further below. A decrease in Orange Book patent district court filings was also seen in 2018 (data from Lex Machina).
The vast majority (more than 90%) of Orange Book patents challenged in IPRs are also involved in district court litigation. And because settlement rates, particularly at the pre-institution stage, are much lower for Orange Book patents (3%) than for IPRs overall (13%), and litigation stays are uncommon, Orange Book patent owners must typically defend their patents in both proceedings simultaneously.
Institution Decision Outcomes for Orange Book Patents
 Most petitions to review Orange Book patents are instituted: two-thirds (66%) of these petitions were instituted on at least one ground through the end of the 2018 fiscal year. (This 66% institution rate is somewhat lower than the 74% institution rate for all IPRs.) Breaking down this data by patent type, compound patents and formulation patents have been the least likely to be instituted on at least one ground (66% and 61% of petitions, respectively), while method of treatment patents were the most likely to be instituted (72% of petitions).
Most petitions to review Orange Book patents are instituted: two-thirds (66%) of these petitions were instituted on at least one ground through the end of the 2018 fiscal year. (This 66% institution rate is somewhat lower than the 74% institution rate for all IPRs.) Breaking down this data by patent type, compound patents and formulation patents have been the least likely to be instituted on at least one ground (66% and 61% of petitions, respectively), while method of treatment patents were the most likely to be instituted (72% of petitions).
While PTAB reviews may be instituted unless the patent owner shows a significant procedural or substantive deficiency with the petition, such as failure to establish the prior art status of a reference (e.g., IPR2014-01126, Paper 22 at 13), failure to address a claim element (e.g., IPR2016-01461, Paper 9 at 10-11), or technical errors (e.g., IPR2014-01329, Paper 33 at 14-15), our results suggest that the claim type may also impact the likelihood of obtaining an institution denial.
The increase in the rate of institution denials is good news for patent owners. Although initial denial rates were low, they rose significantly in 2018, approaching 60%. Patent owners are watching carefully to see whether this trend holds, and whether it signals that the PTAB is becoming friendlier to Orange Book patents or that patent defense strategies are improving (or both).
Final Written Decision Outcomes for Orange Book Patents
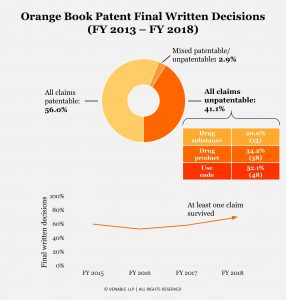 Of all petitions directed to Orange Book patents that reached the final written decision stage, the PTAB found at least one instituted claim patentable in 59% of the cases—a much higher rate than for petitions generally (44% of cases). But there has also been a profound difference between the rates of patent survival across the different Orange Book patent types. At least one instituted claim survived in 80% of proceedings concerning compound patents. By contrast, at least one claim of the formulation and method of treatment patents survived in 66% and 48% of proceedings, respectively. Compound patents held up much better than Orange Book patents overall. Formulation patents fared a bit better than average, and method of treatment patents were the least likely to survive the final written decision stage with at least one claim intact.
Of all petitions directed to Orange Book patents that reached the final written decision stage, the PTAB found at least one instituted claim patentable in 59% of the cases—a much higher rate than for petitions generally (44% of cases). But there has also been a profound difference between the rates of patent survival across the different Orange Book patent types. At least one instituted claim survived in 80% of proceedings concerning compound patents. By contrast, at least one claim of the formulation and method of treatment patents survived in 66% and 48% of proceedings, respectively. Compound patents held up much better than Orange Book patents overall. Formulation patents fared a bit better than average, and method of treatment patents were the least likely to survive the final written decision stage with at least one claim intact.
Unlike the significant change in institution rates over time, the rate of Orange Book petitions with at least one claim surviving the final written decision stage has remained relatively constant, at around 60%, and has been slowly increasing since 2016.
Review of IPR Decisions at the Federal Circuit and Decisions on Remand
We also reviewed the results on appeal and remand of all IPR decisions (not solely directed to Orange Book patents). In its review of IPR decisions, the Federal Circuit has affirmed every issue in nearly three-quarters of cases, showing substantial deference to the PTAB’s conclusions. The Federal Circuit reversed or vacated part or all of the PTAB’s decisions in about 10% and 13% of cases, respectively, and did not reach a decision on the merits in the remaining cases. When remanded from the Federal Circuit, the PTAB kept its original opinion unchanged in more than 80% of cases. Thus, appeals and remands have not been a very successful strategy for changing the PTAB’s initial determination.
Conclusion
Based on our analysis, it appears that the pendulum is swinging back in favor of Orange Book patent owners. The number of petitions challenging these patents has slowed since the high rates seen in the 2015 and 2016 fiscal years. The rate of institution denials of Orange Book patents has risen considerably, and there has been a slow but steady increase in the number of Orange Book patents surviving the final written decision, although success rates vary notably among patents claiming compounds, formulations, and methods of treatment in the Orange Book. It remains to be seen whether these trends continue through 2019.
Methodology
Considerable effort was made to identify all petitions concerning patents that are now or have been listed in the Orange Book. We compiled monthly versions of the Orange Book from 2014 through October 2018 (current version available here). Orange Book listed patents were subclassified by type according to the “Drug Substance” and “Drug Product” flags and Use Codes listed in the Orange Book. Patents with a “Drug Substance” flag were considered to be compound patents. Patents with a “Drug Product” but no “Drug Substance” flag were considered to be formulation patents. Finally, patents with a Use Code but no “Drug Substance” or “Drug Product” flag were considered to be method of use patents.
Joined petitions were counted individually for purposes of analyzing the petition filing and institution rates, and together for the purpose of analyzing final written decision outcomes. Unless otherwise reported, the data presented was analyzed through the end of the USPTO’s 2018 fiscal year, which was September 30, 2018.
In evaluating outcomes at the institution and final written decision stage, we looked only at cases where the Board issued a decision; we did not include cases where the parties settled, the Petitioner abandoned its challenge, or the Patent Owner disclaimed all claims.
Charts Provided by Venable
Image ID: 20566277

![[IPWatchdog Logo]](https://ipwatchdog.com/wp-content/themes/IPWatchdog%20-%202023/assets/images/temp/logo-small@2x.png)


![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Patent-Litigation-Masters-2024-sidebar-early-bird-ends-Apr-21-last-chance-700x500-1.jpg)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/WEBINAR-336-x-280-px.png)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/2021-Patent-Practice-on-Demand-recorded-Feb-2021-336-x-280.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/Ad-4-The-Invent-Patent-System™.png)







Join the Discussion
No comments yet.