Merck & Co. (NYSE: MRK), which is headquartered in Whitehouse Station, NJ, and also does business under the name Merck Sharp & Dohme MSD, is one of the world’s largest and most renowned pharmaceutical development companies. The company just made a major acquisition in the middle of December, investing $8.4 billion in the purchase of Cubist Pharmaceuticals, a deal which has been questioned by those covering the pharmaceutical industry after two of Cubist’s patents protecting the antibiotic pharmaceutical Cubicin were declared invalid in U.S. court. In a much smaller December deal, Merck also paid $375 million to acquire Switzerland-based OncoEthix, a pharmaceutical developer who has a drug pipeline full of cancer treatments. Merck, along with NewLink Genetics Corporation of Ames, IA, was also recently named as a recipient of a $30 million contract from the U.S. Department of Health and Human Services for an Ebola vaccine candidate being jointly developed by those companies.
It’s been a while since we’ve scanned the patent applications filed by Merck with the U.S. Patent and Trademark Office and this latest survey uncovered a great number of innovations in the field of cancer treatments. Most of these involve the use of pharmaceutical compounds which inhibit the activity of certain proteins in the human body which contribute to the spread of different cancers. Other patent applications we explored include treatments for neurological disorders, including Alzheimer’s disease, as well as the hepatitis C virus.
The strong patent portfolio of Merck received many important additions over the past couple of months. Our readers can explore a pair of patents issued to protect pharmaceutical treatments for metabolic disorders, including one that regulates cholesterol levels to reduce the risks of coronary heart disease. Another patent protects a pharmaceutical compound designed to treat or prevent migraines and headaches in patients. We also profile a couple of patents issued to a Netherlands-based subsidiary of the corporation, including an applicator that enables easier implants of contraceptive devices under a patient’s skin.
[Companies-1]
Merck’s Patent Applications: Cancer Treatments as well as Medications for Alzheimer’s, Hepatitis C
Like any pharmaceutical company, research and development is a major investment over at Merck. In 2013, the company invested more than $7 billion in R&D activities; the company’s official website cites statistics indicating that the average cost of developing a single successful medication is about $1 billion, including monies spent developing compounds which fail to become drugs. Along with novel pharmaceutical compounds, Merck is also involved in developing digital solutions for health care through the activities of its Merck Medical Information and Innovation (M2i2) group for establishing partnerships to explore possibilities within that field. In July of last year, the company opened an office in the innovation hub in Philadelphia’s University City Science Center which also focuses on information technologies in health care.
Innovative pharmaceutical compounds that target cancers were far and away the most prevalent new developments reflected in the patent applications filed by Merck with the USPTO over the past few months. The inhibition of specific proteins that contribute to the spread of cancers through their mutation is the focus of U.S. Patent Application No. 20140323519, which is titled Heterocyclic Compounds as B-Raf Inhibitors for Treatment of Cancer. The heterocyclic compound of the specific formula claimed by the patent can be administered to a patient suffering from cancer or other cellular proliferative diseases to inhibit the activity of B-Raf. B-Raf is a protein kinase isoform that mutates at a high frequency in the case of cancers and inhibition of this substance has the potential to block biochemical pathways important to the spread of cancers.
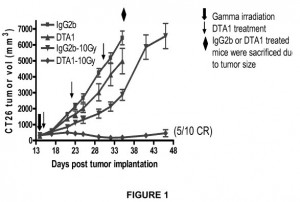 Another cancer therapy which targets the activity of a protein within the human body is discussed within U.S. Patent Application No. 20140348841, filed under the title Anti-GITR Antibodies. This compound is intended to support the activation of glucocortinoid-induced TNFR-related protein (GITR), which increases the resistance of a patient to tumors and viral infections. The binding compound that binds to human GITR claimed by the patent application is comprised of an antibody light chain variable domain and an antibody heavy chain variable domain, each domain having three complementarity domain regions (CDRs) from a specific group.
Another cancer therapy which targets the activity of a protein within the human body is discussed within U.S. Patent Application No. 20140348841, filed under the title Anti-GITR Antibodies. This compound is intended to support the activation of glucocortinoid-induced TNFR-related protein (GITR), which increases the resistance of a patient to tumors and viral infections. The binding compound that binds to human GITR claimed by the patent application is comprised of an antibody light chain variable domain and an antibody heavy chain variable domain, each domain having three complementarity domain regions (CDRs) from a specific group.
Improvements to multi-agent therapies for battling against the spread of certain types of cancer are described by U.S. Patent Application No. 20140343071, which is titled Compositions and Methods for Treating Cancer. The patent application would protect a method of treating certain types of cancers, including breast, colorectal and ovarian cancers, through the use of both a WEE1 inhibitor and a CHK1 inhibitor. This drug combination is designed to inhibit both CHK1, a serine/threonine kinase that can contribute to aberrant DNA replication, as well as WEE1, a tyrosine kinase which can be interfered with to repress cancer cell proliferation. The antagonizing of certain proteins to treat or prevent cancers is at the center of U.S. Patent Application No. 20140357618, filed under the title Substituted Imidazopyridines as HDM2 Inhibitors. The compound treats cancers by inhibiting the activity of Human Double Minute 2 (HDM2) proteins, which act as a regulator of p53, a tumor suppressing protein which maintains the integrity of the genome in a cell by regulating gene expressions related to DNA repair and growth arrest.
We found other treatments developed by Merck that are designed to treat infections involving the hepatitis C virus, which can cause a range of chronic liver disease in an individual. U.S. Patent Application No. 20140371138, which is titled Inhibitors of Hepatitis C Virus Replication, would protect a treatment for the hepatitis C viral replication in a patient through the inhibition of the zinc metalloprotein known as NS5A. NS5A, or nonstructural 5A protein, interacts with numerous host-signaling pathways which have suggested to researchers that this particular zinc metalloprotein may modify a host cell environment so that the cell enters a state where it is more susceptible to the virus.
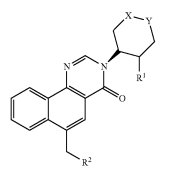 The treatment of Alzheimer’s disease and other neurological conditions may become possible through the compound described within U.S. Patent Application No. 20140364395, titled Pyranyl Aryl Methyl Benzoquinolinone M1 Receptor Positive Allosteric Modulators. The pharmaceutical compound claimed by the patent has the capability to modulate the muscarinic M1 receptor, which is prevalent in a person’s hippocampus, striatum and cerebral cortex and plays a major role in cognitive processing. Aside from Alzheimer’s disease, disorders which may be treated by altering the activity of the M1 receptor include pain disorders, sleep disorders or schizophrenia.
The treatment of Alzheimer’s disease and other neurological conditions may become possible through the compound described within U.S. Patent Application No. 20140364395, titled Pyranyl Aryl Methyl Benzoquinolinone M1 Receptor Positive Allosteric Modulators. The pharmaceutical compound claimed by the patent has the capability to modulate the muscarinic M1 receptor, which is prevalent in a person’s hippocampus, striatum and cerebral cortex and plays a major role in cognitive processing. Aside from Alzheimer’s disease, disorders which may be treated by altering the activity of the M1 receptor include pain disorders, sleep disorders or schizophrenia.
We also noted an intriguing Merck pharmaceutical innovation which is aimed at producing more effective vaccines for preventing influenza in individuals. U.S. Patent Application No. 20140314797, titled Yeast Strain for the Production of Proteins With Terminal Alpha-1,3-Linked Galactose, claims a yeast or filamentous fungus host cell capable of producing recombinant glycoproteins having N-glycans with a specific predominant terminal sugar structure. The genetically engineered yeast strains of this invention can be used in the production of vaccines that have increased immunogenicity by eliciting an elevated immune response.
Issued Patents of Note: From Surgical Implant Applicators to Metabolic Disorder Treatments
Of the pharmaceutical corporations included in the Companies We Follow series, Merck has been building one of the stronger patent portfolios that we’ve surveyed in recent years. Our 2013 U.S. patent total statistics put together by the Intellectual Property Owners Association reports that this company was 126th overall that year with 268 patents issued by the USPTO. This represented a reduction in total patents awarded to the company in the previous year by only about three percent.
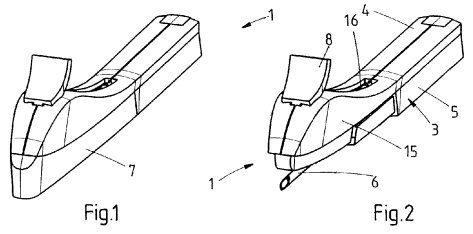 We noted a couple of novel innovations protected for a Netherlands-based subsidiary of Merck, known as Merck Sharp and Dohme B.V., meant to improve the health or affect the operation of the human reproductive system in patients. U.S. Patent No. 8888745, issued under the title Applicator for Inserting an Implant, claims an applicator for inserting an implant under the skin that has a housing, a cannula with an insertion length extending outside of the housing and a handle for grasping and maneuvering the applicator and cannula during the insertion of an implant. This applicator enables a surgeon to implant a rod-like contraceptive implant by gently lifting the skin in a way that reduces the risk of causing distress to a patient.
We noted a couple of novel innovations protected for a Netherlands-based subsidiary of Merck, known as Merck Sharp and Dohme B.V., meant to improve the health or affect the operation of the human reproductive system in patients. U.S. Patent No. 8888745, issued under the title Applicator for Inserting an Implant, claims an applicator for inserting an implant under the skin that has a housing, a cannula with an insertion length extending outside of the housing and a handle for grasping and maneuvering the applicator and cannula during the insertion of an implant. This applicator enables a surgeon to implant a rod-like contraceptive implant by gently lifting the skin in a way that reduces the risk of causing distress to a patient.
An intra-vaginal device for the administering of active agents to female patients is disclosed and protected by U.S. Patent No. 8900615, which is titled Drug Delivery System. It claims a vaginal ring having a drug-loaded ethylene-vinylacetate copolymer core, an intermediate layer composed of the same and a non-medicated ethylene-vinylacetate copolymer skin which covers the intermediate layer. This device supports the simultaneous release of two or more active substances for various medical purposes, including contraception and hormone replacement.
A couple of patents issued in recent months to Merck protect medications which have been developed to treat a range of metabolic disorders and conditions. A weight loss treatment that targets obesity-related and diabetes-related disorders in patients is the focus of U.S. Patent No. 8921394, issued under the title Prolylcarboxypeptidase Inhibitors. The pharmaceutical compound is designed to improve upon the weight loss efficacy of conventional drugs, which can bring significant side effects, and bariatric surgeries, which results in an increased mortality rate within a year of surgeries. The inhibition of the serine protease enzyme prolylcarboxypeptidase, or PRCP, can help to decrease food intake in patients who are administered the treatment. The metabolic control of lipoprotein levels, which combine with and transport fats and other lipids through the blood, is the goal of the pharmaceutical compound protected by U.S. Patent No. 8865707, which is titled Cholesteryl Ester Transfer Protein Inhibitors. This compound works to inhibit the activity of cholesteryl ester transfer protein, or CETP, a protein which is not present in animals that are resistant to coronary heart disease. The inhibition of CETP in humans is intended to raise levels of high density lipoprotein cholesterol (HDL-C) and reduce levels of low density lipoprotein (LDL-C), both of which are associated with a lower risk of coronary heart disease.
Those who suffer from migraines and intense headaches may find some relief from the Merck innovation outlined by U.S. Patent No. 8912210, entitled Piperidinone Carboxamide Azaindane CGRP Receptor Antagonists. The pharmaceutical compound claimed here works to provide antagonism of calcitonin gene-related peptide, or CGRP, to help treat a number of pain and inflammatory disorders for which CGRP plays an important role in neuromodulation. The pharmaceutical compound of this invention can be used to treat a migraine condition after it has started or be used for its prophylactic effect in preventing migraines and related conditions from occurring.
We’ll close our coverage of Merck’s recently patented technologies with a closer look at an innovation which supports the sustained release of a variety of pharmaceutical treatments to increase the beneficial effects of those treatments within a patient’s body. U.S. Patent No. 8852632, entitled Pharmaceutical Formulation Containing a Release Rate Controlling Composition, claims a pharmaceutical composition for solid dose oral administration which contains an effective amount of a potassium salt of a compound as well as a release rate controlling composition which is made up of a solubilizing agent, a gelling agent and a water soluble filler, which is optional. This invention is intended to delay the rapid absorption of a solid dosage compound for greater bioavailability of the compound within the stomach and the gastrointestinal tract of a patient.

![[IPWatchdog Logo]](https://ipwatchdog.com/wp-content/themes/IPWatchdog%20-%202023/assets/images/temp/logo-small@2x.png)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Patent-Litigation-Masters-2024-sidebar-early-bird-ends-Apr-21-last-chance-700x500-1.jpg)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/WEBINAR-336-x-280-px.png)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/2021-Patent-Practice-on-Demand-recorded-Feb-2021-336-x-280.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/Ad-4-The-Invent-Patent-System™.png)







Join the Discussion
No comments yet.