Seemingly every new court decision addressing subject matter eligibility under 35 U.S.C. § 101 progressively weakens the patent system, especially in the life sciences. And each case seems to present a new low in terms of the depth and quality of analysis. Courts have misapplied § 101 in a variety of contexts against a wide range of technologies.
But I perceive a common thread running through these decisions, typified in the Federal Circuit’s recent decision in Ariosa v. Sequenom, supporting the growing feeling amongst the innovation community that § 101 decisions have taken on an arbitrary, outcome-driven quality warned against by Judge Moore in the AMP v. Myriad case:
Holding isolated DNA not patentable would destroy long settled industry expectations for no reason other than a gut feeling that DNA is too close to nature to be patentable, an arbitrary decision based on a judge-made exception.
A close analysis of recent court decisions shows how early in the process the ultimate outcome is set. Courts are striking down patents simply because the challenger has convinced the judge(s), on Judge Moore’s “gut” level, that the patents are bad, e.g., for patients, for large electronics companies supposedly pestered by trolls, etc. The first, and practically speaking last, step in the process of reaching a policy-driven decision to arbitrarily strike down an unpopular patent is to broadly and imprecisely define what is claimed in terms of its “essence.”
Essence of the Claim
The first and most critical task in most eligibility analysis is carefully and precisely defining the subject matter of the claims as well as the natural phenomenon that is potentially being claimed. The superficial definitions in current court decisions are usually found in the part of a decision where the court explains what the patentee “essentially” or “generally” claimed. I understand that judges are not technology experts, but the current shortcut of boiling claims down to their “essence” is particularly problematic in a § 101 case. Everything is a “natural phenomenon” if this term is treated expansively. Easier to define (and thus more appropriate as a ground for objective rather than arbitrary eligibility analysis) are “products of nature” and “natural laws.” Unfortunately, patent examiners and courts have jumped to “natural phenomena” as their go-to basis for § 101 rejections precisely because it is amorphous and may be used to readily reject or invalidate a claim they don’t like.
Hence the majority’s statement of the case:
Thus, the claims at issue, as informed by the specification, are generally directed to detecting the presence of a naturally occurring thing or a natural phenomenon, cffDNA in maternal plasma or serum. As we noted above, the claimed method begins and ends with a naturally occurring phenomenon.
Others have appropriately criticized the lack of depth of analysis typified by this ill-defined “begins and ends” test. Here I focus more on the problems with choosing such a generalized and imprecise starting point, and how such a starting point all but guarantees an outcome of invalidity.
The natural product in Ariosa was DNA, not “cffDNA.” I think Sequenom was seriously harmed in this case by what I call the jargon jinx. People love jargon and snappy acronyms, especially if that jargon is coined incident to a relatively new, significant scientific advance. Pursuant to this general human tendency, “cffDNA” in this case gained a special status and somehow came to be regarded as some special new type of DNA. It is not; it is DNA. The “natural product” at issue in this case was DNA. If we must get a little more specific, then we can say the judicial exception in this case was fetal DNA. The fact that fetal DNA is found in various locations may be a “natural phenomenon” in the broadest sense of that term, but that is not a judicial exception (natural product or natural law) that is even capable of claiming. This is particularly tragic when you consider the claims themselves never use the term “cffDNA.” They instead say “a paternally inherited nucleic acid of fetal origin [in] a maternal serum or plasma sample.”
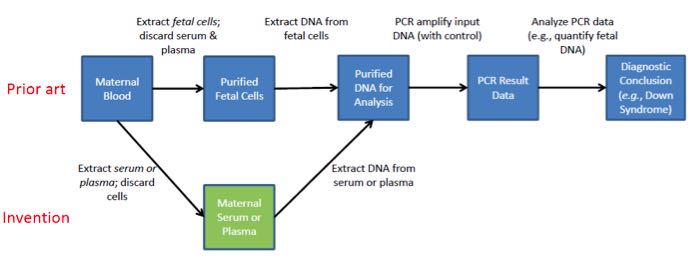
To illustrate the importance of the difference between claims to DNA and Sequenom’s claims to a laboratory method of processing samples for DNA testing, let’s suppose the claim read “A fetal DNA molecule located in maternal serum or plasma” or even “A fetal DNA molecule isolated from maternal serum or plasma.” Such a claim would be ineligible because it claims fetal DNA (which is a natural product), not because it claims the “natural phenomenon” of fetal DNA being located in maternal serum. The patent challenger successfully caught the court up in the drum beat of “Sequenom is claiming cffDNA” when Sequenom did nothing of the sort.
The record suggests the presence of certain fetal biomolecules (e.g., proteins) in maternal serum was well-known at the time of filing. The same is true for the presence of “cell-free” DNA of other origin (e.g., from tumors). Most importantly, however, Sequenom’s patent itself teaches it was known in the art that fetal cells can pass into the mother’s blood. Diagnostic techniques had been devised to isolate these cells and analyze the fetal DNA extracted from them, but these techniques were expensive and time consuming. When viewed from this background, the claimed invention is clearly a technical improvement in the processing of samples for prenatal diagnosis.
Correctly Envisioning the Invention
The summary of the invention in the patent begins as follows: “This invention provides a detection method performed on a maternal serum or plasma sample from a pregnant female, which method comprises detecting the presence of a nucleic acid of foetal origin in the sample.” Thus, the invention in Ariosa lies in devising a new sample input for a laboratory process, thus greatly improving that process. Sequenom did not invent or claim fetal DNA. Nor did Sequenom claim all methods of testing fetal DNA or even all methods of testing fetal DNA in maternal blood. Sequenom instead claimed a modification to routine laboratory testing methods by altering how the samples were processed so that fetal DNA testing could be improved.
The following diagram helps illustrate this critical point:
Thus the claim is not to any DNA per se or even the fact of fetal DNA floating around free of cells in maternal plasma (the dangerous jargon of “cffDNA”). It is to an inventive modification in an existing technological process that significantly improves the technical performance of that process. Sequenom failed to get the courts to view the invention from this technological process perspective. Instead the court got hung up on the presence of DNA in the claim and Ariosa’s framing of the case in terms of the location of fetal DNA.
Not that we can fault Sequenom for this failure. Patentees so far have a very poor record in getting judges to see past the rhetoric and understand sometimes complex technical issues (see, e.g., Justice Scalia’s concurrence in AMP v. Myriad). And patent challengers, empowered by convincing PR spin and misdirection about harm to patients, have found all too sympathetic judicial audiences. Courts these days are making policy-based decisions, untethered from any rule of law, aimed at killing patents they don’t like and I am skeptical that any amount of skill could have changed that in Ariosa (or in AMP, In re Roslin, UURF v. Ambry, etc.).
Conclusion
In what other field would devising a new starting material for a process that significantly improves the process’s yield not be considered patentable? It is only when one starts by defining the potentially claimed judicial exception imprecisely and overly broadly (“the natural phenomenon of cffDNA”) that one jumps onto the slippery slope leading to an abyss where nothing in the life sciences is patent eligible. This initial definition of what is claimed and what is the judicial exception against which it must be compared is the fundamental error in most eligibility analyses and the battleground where eligibility will most often be decided. As long as challengers enjoy judges more than willing to adopt their boiled-down, incomplete depiction of claims, I fear the tide will continue to roll against incentives for innovation in the life sciences.

![[IPWatchdog Logo]](https://ipwatchdog.com/wp-content/themes/IPWatchdog%20-%202023/assets/images/temp/logo-small@2x.png)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Artificial-Intelligence-2024-REPLAY-sidebar-700x500-corrected.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/UnitedLex-May-2-2024-sidebar-700x500-1.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Patent-Litigation-Masters-2024-sidebar-700x500-1.jpg)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/WEBINAR-336-x-280-px.png)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/2021-Patent-Practice-on-Demand-recorded-Feb-2021-336-x-280.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/Ad-4-The-Invent-Patent-System™.png)






Join the Discussion
34 comments so far.
Anon
August 13, 2015 07:34 amstep back,
We will have to agree to disagree, as my characterization of spin is absolutely (and sadly) correct.
Spin “works” because as you point out we are not rational creatures.
But that only proves my point.
Spin is not an “excuse”
Spin is an observation of what is going on, by people with vested interests in achieving a certain ends, no matter the means.
I would venture that the frustration is not limited to “techno-geeks.” Instead I would venture that the greater frustration occurs to those that know and understand history and the law, and more specifically the rule of law and the history of innovation.
And even more frustration when the knowledge of “who we are” that transcends “who we think we are” is “politely” ignored and the spin of “say it often enough” is allowed, permitted, and in certain rather obvious fora, encouraged.
step back
August 12, 2015 01:42 pmAnon @30
It’s not “spin”.
It’s how the human brain functions.
Unfortunately we are not rational creatures.
All the evidence around you screams that truth –racism, shootings, witch hunts, stampedes, beheadings, Trump towering above all other candidates and Mount Olympus Nine deciding that BRCA1 molecule is more like plucking a leaf from a tree than lathing the baseball bat from its trunk.
I have been studying the issue of human irrationality for many years, not just for this moment of flash of insight. It’s not something you can wrap your head around with a one word excuse like “spin”.
Yes it is frustrating to techno-geeks such as ourselves. But it is how our species mates operate. They operate by subconscious feelings, not by rational neo-cortical thought process. A sobering and disturbing conclusion. We are not what we think we are.
Paul Cole
August 12, 2015 09:19 amMore @ Anon
Do you REALLY believe that constructive patent reform is possible through Congress and if so how? Do you really believe that US lawmakers would be willing to strip the Supreme Court of its jurisdiction in patent appeals? How might this be achieved?
Or is there a way to reach out to the Justices and some of the Federal Circuit judges and persuade them towards a more patent-friendly attitude? How might this be done?
Paul Cole
August 12, 2015 08:53 am@ Anon
Increasingly we see opinion led by ill-informed opinion formers, and an increasingly divided political process, against which informed debate is difficult. So while not giving up on persuasion, logic and attention to detail, I have come to share your belief that something different is also needed. Do you have any positive suggestions or leads?
Anon
August 12, 2015 07:35 amstep back,
You can lead a horse to water…
More spin is not a really good answer to the problem of too much spin in the system already.
step back
August 12, 2015 01:10 amAnon,
Courts are not things. Courts are people. Different people at different courts.
People will listen to a message if it resonates with one of their current belief systems.
The problem is how to frame your message so that is in tune with one of the belief systems of a given judge.
Anon
August 12, 2015 12:14 amMr. Cole,
With all due respect, you keep on advocating the wrong thing.
The courts just are not listening.
We need a different solution.
A Rational Person
August 11, 2015 05:42 pmstep back @24 One of the things that is interesting to me about Frankfurter’s concurring opinion is that it includes a dead-on criticism of one of the reasons why the Prometheus and Mayo are such bad decisions, i.e., they create a standard so subjective and vague that it could be used to invalidate any patent.
Even though technology and what we now know about the world, Frankfurter’s basic reasoning, unlike the reasoning in the majority’s opinion, is still sound and would still be sound under 35 USC 101 which postdates the Funk Bros. decision. In fact, had 35 USC 112 existed at the time of Funk Bros., based on Frankfurter’s reasoning, I think he would have argued that the problem with the claims at issue was a 35 USC 112 enablement problem, not a 35 USC 101 patent eligibility problem.
step back
August 11, 2015 05:25 pmDate of Funk Bros. =1948
Date of more modern Patent Statute= 1952
https://en.wikipedia.org/wiki/Funk_Bros._Seed_Co._v._Kalo_Inoculant_Co.
Paul Cole
August 11, 2015 04:59 pm@ Step Back
It is an unavoidable feature of the law that old decisions have modern consequences. Decisions like Funk cannot be avoided, only understood correctly and placed in their proper context.
step back
August 11, 2015 04:36 pmPaul and Rational,
I think it does a great disservice to modern patent practice to give deference to judicial opinions penned in days gone by, before scientists better understood the workings of the universe and our place within it as irrational freak outcomes of random biological evolution.
I would prefer the explanations in opening chapters of Roger Penrose’s “Road to Reality” where he explains that our ideas (e.g. abstract mathematical ideas) of how the universe is put together are likely to be wrong, but are generally good enough for modeling sake. For example, Euclidean geometry “assumes” continuity and consistent linearity from here to infinity and beyond. Quantum mechanics informs us though that space is not continuous and different laws appear to apply at different scales of distance (subatomic versus galactic). So we are incorrect from the get go.
All these so-called “laws of nature” are actually man-made abstract ideas about how nature works. To say that one may not patent either abstract ideas “or” laws of nature constitutes mere redundancy, not deep thinking.
There is a fundamental difference between claiming exclusive rights to abstract ideas themselves and to claiming exclusive rights in new and useful machines, compositions of matter, etc. whose usefulness can be explained in terms of abstract ideas (e.g. with aid of mathematical formula –even if Euclidean math is wrong).
Our Mount Olympus 9 can’t grasp that simple concept and there lies their befuddlement. Just because they cannot comprehend it, that means to them that no one should be allowed to patent the latter. It is not inventors and their devious patent attorneys (with use of draftsman’s trickery) who are holding back innovation but rather the Medieval-wise cloaked denizens of the high bench.
Paul Cole
August 11, 2015 03:27 pm@ A Rational Person
I have spent many tens of hours trying to gain a fuller understanding of Funk Brothers, including investigation online of the biographies of Justice Douglas and Justice Frankfurter.
Owing to the importance of this case for modern day issues it has been inadequately researched. Justice Douglas tended to write relatively cursory opinions, according to his biographers because he had been passed over for the 1944 vice-presidential nomination. Justice Frankfurter, also according to his biographers had an argumentative style that was not popular with his colleagues. Some of the internal conflict within the Court may have been responsible for the preference for the Douglas opinion over the more conservative and better reasoned Frankfurter opinion. It would be a service to our profession if a public-spirited Washington-based firm arranged for inspection of the file and made accessible online the relevant documents for that case, including the Supreme Court briefs and any internal documents recording the discussions between the justices.
The Douglas opinion is very difficult to interpret and modern day judges have not attempted to do so, but merely cited isolated passages out of their original context and with scant attention to their original meaning and intention. Funk Brothers has been cited as an eligibility case, ignoring the inconvenient truth that much of the reasoning of Justice Douglas was based on his Cuno opinion, and that automobile cigarette lighters do not grow on trees.My own interpretation is to be found on Patent Docs, but what I have been able to do is limited by the availability of this additional background material.
http://www.patentdocs.org/2015/04/gues-post-microorganisms-funk-brothers-chakrabarty-and-trips.html
A Rational Person
August 11, 2015 02:52 pmstep back@20,
I forgot to mention that at least one former Supreme Court justice, Felix Frankfurter, may have been a super braniac with respect to the fields of chemistry and biology of his day. Unlike the majority opinion in Funk Bros., Frankfurter’s concurring opinion shows that he understood both the patent law-related issues and the technical issues involved in the case.
In particular, I am impressed with Frankfurter’s prescience in this passage from his concurring opinion:
“It only confuses the issue, however, to introduce such terms as ‘the work of nature’ and the ‘laws of nature.’ For these are vague and malleable terms infected with too much ambiguity and equivocation. Everything that happens may be deemed ‘the work of nature,’ and any patentable composite exemplifies in its properties ‘the laws of nature.’ Arguments drawn from such terms
for ascertaining patentability could fairly be employed to challenge almost every patent. On the other hand, the suggestion that ‘if there is to be invention from such a discovery, it must come from the application of the law of nature to a new and useful end’ may readily validate Bond’s claim. Nor can it be contended that there was no invention because the composite has no new
properties other than its ingredients in isolation. Bond’s mixture does in fact have the new property of multi-service applicability. Multi-purpose tools, multivalent vaccines, vitamin complex composites, are examples of complexes whose sole new property is the conjunction of the properties of their components. Surely the Court does not mean unwittingly to pass on the patentability of such products by formulating criteria by which future issues of patentability may be prejudged. In finding Bond’s patent invalid I have tried to avoid a formulation which, while it
would in fact justify bond’s patent, would lay the basis for denying patentability to a large area within existing patent legislation.”
A Rational Person
August 11, 2015 02:14 pmstep back@20 very well said. I agree 100%.
step back
August 11, 2015 01:04 pmRational@19
Actually we “human” animals are highly irrational.
A good litigator knows that and plays to worst in our animal instincts.
So to expect that the “truth” will come out during litigation is plain nuts.
The truth is that judges are not super brainiacs and they will not learn molecular biochemistry through one hour of tutoring when it takes many college level semester hours for the best and the brightest among our youth to learn and understand that stuff.
Judges can have huge egos. And therefore when a manipulative litigator stokes those egos by telling them they understand a biochemistry concept as being analogous to plucking a leaf from a tree or lathing a baseball bat out of its trunk they can feel mighty proud of their comprehension abilities when in fact their comprehension is zero. I’m talking about the oral arguments during the SCOTUS Myriad v ACLU case of course. To not understand that a DNA fragment is a separate and different molecule (a new and useful composition of matter) from a full DNA strand is to fail miserably at organic chemistry 101. That is precisely what the Mount Olympus 9 did in Myriad and they have the chutzpah to be proud of their failing workproduct. Welcome to our real world version of that movie, Idiocacy. Brando, it’s got electrolytes.
http://patentu.blogspot.com/2014/12/discovery-death-squads.html
A Rational Person
August 11, 2015 11:29 am“The second, exemplified by Ariosa is an inability to explain the technical facts to inexperienced judges. As a profession we need to brush up our standards of advocacy, and also of patent drafting.”
What if the “inexperienced judges” have a poor understanding of biology and don’t care if they don’t understand the technical facts? What good is a lawyer explaining the technical facts to the inexperienced judges if the judges don’t want to spend the time to learn about the technical facts? As the saying goes, “You can lead a horse to water . . . .”
For example, see Justice Scalia’s concurring opinion in Myriad. At least he was honest about his ignorance of the “fine details of molecular biology.”
Paul Cole
August 11, 2015 07:26 amIn a posting on the Patent Docs blog I pointed out that the factual error in this case is quantifiable – to say that the PCR amplified sequence is a natural rather than a synthetic product is in error by a factor of 10exp3 to 10exp6 on the face of the opinion of Judge Reyna. That is a wide enough margin to amuse anyone. See
http://www.patentdocs.org/2015/07/news-from-abroad-ariosa-diagnostics-v-sequenom-and-isis-innovation-a-european-view.html
I think a number of commentators have been making essentially the same point here.
In truth you can, I think, pull the invention out of the Ariosa claim. But it is decidedly broad and woolly, and the sad truth is that claims of that kind end up in trouble, especially with judges who do not have a long history of experience in patent matters.
There are two major problems with Section 101 cases. The first is over-broad lawyer cringe in the face of Supreme Court opinions. Due deference to the Supreme Court, I submit, involves precisely the same “briefing” analysis as for any other court opinion. If that is done, then recent Supreme Court opinions are decidedly narrower than currently believed in our profession. It does no service to the Supreme Court to misinterpret its opinions, and that must be true irrespective of whether the erroneous interpretation is of undue breadth or inadequate breadth. The level of interpretation at the USPTO and in cases such as Roslin and Ariosa would be within the reach of a bright contributor to the New York Times (or this week The Economist – ugh!) with a degree in economics, but should earn a failing grade at any self-respecting law school. The second, exemplified by Ariosa is an inability to explain the technical facts to inexperienced judges. As a profession we need to brush up our standards of advocacy, and also of patent drafting.
step back
August 10, 2015 03:22 pmRational @16
I think we live in a country /world where everybody kicks the can forward in hopes that the next person will take care of it.
I have no idea if any of the Fed. Cir. judges understand how irrational and outside the bounds of Congressional law stand the Alice, (Bilski) Myriad and Prometheus decisions.
If they do understand, then they are kicking the can forward to Congress to fix the problem. But Congress now has its attentions fixated on the 3 ring circus acts for the 2016 Presidential election. It may not be until 2017 before Congress can even start “directing” its ADHD attentions to the plight of US inventors. By then however, Congress may be bought and owned by them that don’t like inventors and patents too much. So it will be too late. Sad. 🙁
A Rational Person
August 10, 2015 02:57 pmStep Back@14
I wish we lived in a country where the Federal Circuit judges had enough guts to say with respect to the Alice, Myriad and Prometheus decisions that because: (1) judicial exceptions to a statute must be interpreted narrowly to avoid having the exceptions swallow the rule, (2) the Supreme Court has provided the Federal Circuit with no definition of terms such as “abstract”, “laws of nature”, “conventional”, etc. and (3) a Court applying the rulings in Alice, Myriad and Prometheus broadly could potentially invalidate any patent, the Federal Circuit, will only apply the rulings of Alice, Myriad and Prometheus to patent claims that are clearly analogous to the claims considered in those cases.
Instead, we appear to live in the land of Oz.
step back
August 10, 2015 01:50 pmAnon @10
Good literary analogy (to wizard of Oz).
Our country and courts have come to be populated by Munchkin Minded little kids (Lollipop Kids) instead of grown rational adults.
It appears to be beyond the mental reach of the Mount Olympus Nine that we send our kids to universities for many years of study to acquire PhDs in biochemistry and such because it is complex and hard to understand stuff; not grasped in a one day tutorial. The Mount Olympus inquisition panels engage in magical Wizard of Oz thinking when they jiggery pokery elevate themselves to being technologically savvy equals when it comes to matters of biochemical and information technology matters.
step back
August 10, 2015 01:40 pmRational Person@13
The other point to add to this discussion is that the US Supreme Court is, under our Constitution, basically above jurisdictional limits put into place by Congress for all “inferior” Article III and Article I tribunals.
The Fed. Cir. is an Article III court subject to the Congress-passed laws that created it. It does not have the jurisdictional authority to ignore Congress-passed laws such as 35 USC 101, 102 and 103. 35 USC 102 says a person “shall” be entitled to a patent unless …. The “unless” part does not include a passage saying unless SCOTUS makes up its own new and irrational legislation regarding patent eligibility.
That is why the Sequenom decision is so troublesome.
A Rational Person
August 10, 2015 11:42 amFNate, from your post @9:
“Hence, my comment. For me, the critical paragraph in the decision appears on page 11 of the authorised report:
‘Using methods like PCR to amplify and detect cffDNA was well-understood, routine, and conventional activity in 1997. The method at issue here amounts to a general instruction to doctors to apply routine, conventional techniques when seeking to detect cffDNA. Because the method steps were well-understood, conventional and routine, the method of detecting paternally inherited cffDNA is not new and useful. The only subject matter new and useful as of the date of the application was the discovery of the presence of cffDNA in maternal plasma or serum.’”
35 USC 101 reads: Whoever invents or discovers any new and useful process, machine, manufacture, or composition of matter, or any new and useful improvement thereof, may obtain a patent therefor, subject to the conditions and requirements of this title.
So it appears that you approve of the Court completely ignoring the word “discovers” out of 35 USC 101? If you feel that the Court did not ignore the word “discovers” in 35 USC 101, how do you explain the following sentence from the opinion: “The only subject matter new and useful as of the date of the application was the discovery of the presence of cffDNA in maternal plasma or serum.’”
Would you approve of Courts in general ignoring other words in statutes or even the constitution? For example, for the sake of the “public good” would you approve of reading out the word “just” from the phrase “just compensation” in the 5th amendment, so the government could grab land for new parks, new highways, new industrial parks, etc. by paying the previous owners of the land just a few dollars, which would be “compensation” but not “just compensation”?
Curious
August 10, 2015 11:08 amThe means to any specific end really do matter.
A point so very few realize when it comes to the law. Too many people think … “well, the judge got this decision correct” while ignoring how the judge got there. If the judge trampled on certain individual’s rights to arrive at the so-called “correct” decision, then it was a bad decision.
The same people who applaud this outcome-driven decision making are the same people who don’t understand why the judges screwed up so badly when the same outcome-driven decision making is used for a decision they do not like. They’ll say “the judge ignored both the facts and the law to arrive at this decision.” However, this is what you get when you elevate the ends over the means. The means do matter.
If this invention was obvious, then 103 was the proper section to use.
Anon
August 10, 2015 07:09 amNate,
You continue to miss the point that the law is not being used properly.
The means to any specific end really do matter.
Anon
August 10, 2015 07:06 amThe truly sad thing about all the “jiggery pokery” is that it breeds a level of purposeful un-critical thinking.
There is much then (and sadly so) to the comparisons that can be made with the Wizard of Oz, pushing dials and spinning wheels behind the curtain.
“You people should consider yourselves lucky that I’m granting you an audience tomorrow instead of 20 years from now.”
“ I’ll get you, my pretty, and your little dog, too!” – If that little dog had been “gotten,” then the curtain would not have been pulled back, and the audience “tomorrow” would have been turned into the eventual audience twenty years hence.
I will repeat again: it is high time our Congress remove the “Wizard” from (re-)writing patent law.
Nate Browne
August 10, 2015 07:01 amYes, I understand that the patent was directed to the detection of patent illegible material (the cell free DNA). And, yes, the cell free DNA was a discovery made by Sequenom.
However, the patent itself was for the “methods” of detection (PCR) which were, and are, routine and obvious to anyone knowledgable in the art.
Hence, my comment. For me, the critical paragraph in the decision appears on page 11 of the authorised report:
‘Using methods like PCR to amplify and detect cffDNA was well-understood, routine,
and conventional activity in 1997. The method at issue here amounts to a general instruction to doctors to apply routine, conventional techniques when seeking to detect
cffDNA. Because the method steps were well-understood, conventional and routine, the method of detecting paternally inherited cffDNA is not new and useful. The only
subject matter new and useful as of the date of the application was the discovery of the presence of cffDNA in maternal plasma or serum.’
I am very impressed that the Court of Appeals could not be hoodwinked by Sequenom’s undoubtedly very expensive lawyers.
But, I know that some of you will want to have these judges hung, drawn and quartered.
step back
August 10, 2015 05:54 amSorry. “know nothing”.
step back
August 10, 2015 05:53 amNate@5,
Ditto what Curious says at 6.
Additionally, it appears from the fact pattern that no one in the prior art had recognized (discovered) that there are tiny amounts of fetal genetic maternal in the maternal blood serum. Without recognition, you cannot have obviousness.
The true mischief of the Sequenom decision and others like it (e.g. Myriad) is that you have no nothing judges pontificating about “nature”, “natural laws” and the word “natural” itself. All these are BS words that have been developed for placating the lay public and then convincing them to buy asparagus water at some big chain supermarkets.
If the Mount Olympus Nine truly understood science, they would know that Mother Nature does not come down from the mountain top with a set of inscribed tablets declaring her top ten “laws”. The things we refer to as Laws of Nature are man-made abstract ideas about how we think nature works. They are not “her” laws. (Often we are wrong in our man-made theories.) Nature quite frankly, is not a nurturing female primate who cares a hoot about us and our prosperity or how accurately we theorize about Nature’s workings. If we go extinct or return the Dark Ages, all the same to Nature. Just one more nuisance infestation on the 3rd rock orbiting the local star.
There is no “natural phenomenon” by way of which maternal blood is sampled, filtered, amplified, etc. to read the tiny amounts of fetal genetic material in it. All that is part of the works and ingenuity of man under the Sun and clearly physical “process” under 101. To rule otherwise is to engage in jiggery pokery and magical thinking.
http://patentu.blogspot.com/2015/06/jiggery-pokery-mock.html
Curious
August 9, 2015 09:37 pmSequenom had claimed non-novel, obvious methods.
If so, then it should have been a rejection under 102/103 — not 101. I suggest you (or anybody) read each section and compare it to the other. They are not the same — not even close.
Personally, I do not care about this technology or any of the parties involved. I just care that the judges applied the proper sections of the law correctly.
You comments about “the US Court of Appeals quite deftly arrested the mischief that would have been created if Sequenom had been allowed their patent” reflects the exact problem identified within the title (i.e., “Outcome-Driven § 101 Decisions”) of this article. The Court should not be monkeying with the law and/or facts to arrive at the outcome they desire. This is how bad law gets made (see, e.g., Benson).
Nate Browne
August 9, 2015 09:29 pmMuch has been written about this decision but nothing quite as misleading as this analysis.
Sequenom had claimed non-novel, obvious methods. In fact, the methods are routinely used in labs and hospitals throughout the world. This case has nothing to do with DNA or cell free fetal DNA (which are the one and the same thing). And, none of this was denied by Sequenom.
In affirming the decision handed down by the US District Court, the US Court of Appeals quite deftly arrested the mischief that would have been created if Sequenom had been allowed their patent.
step back
August 9, 2015 04:19 pmDoug,
I think you will get great resistance from US practitioners to the idea of using Jepson style claims which implicitly include admitted prior art.
Take a look at the new July 2015 PTO guidelines.
I suggest that the PTO itself is starting to question its own right to engage in creative designation of ineligible subject matter categories.
Doug Gilmore
August 9, 2015 11:11 amHas anyone had experience with Jepson claims for trying to overcome §101 rejections in prosecution of biotech, CBM, or software apps? e.g., in the above case, something like:
In a [generally described conventional method for processing a maternal blood sample in preparation for fetal DNA analysis], the improvement comprising the steps of: [description of isolating at least one of serum or plasma from the maternal blood sample; and extracting DNA therefrom prior to PCR amplification].
My interest is more on the software/CBM side of things. Can Jepson claims be used to effectively focus the Examiner’s attention on what constitutes, “significantly more than the judicial exception?”
I understand the dangers of using Jepson claims, but where a §101 has achieved immovable traction, has anyone successfully used them to advance substantive examination?
Anon
August 9, 2015 10:07 amThe “arbitrary” nature runs so deep that even if we allow the Court to invade the domain of the Legislative Branch and re-write patent law (as it is rather obvious that they are so doing), the law so written fails the Void for Vagueness doctrine.
That doctrine is usually applied in criminal manners, but not exclusively so. In civil matters where rights and property are at stake, where a level of fundamental certainty is required a priori, the doctrine comes into play.
And yet another reason why critical thinking – legal critical thinking – needs to highlight just how off the track our jurisprudence on patent law has become because of our High Court.
I had fervently hoped that Alice would have been a case like Diehr and Chakrabarty to restore balance to an anti-patent animus in the Court, but instead the pendulum simply swung even further off-center.
Congress reacted strongly to an anti-patent Court with the 1952 Act. This Court has shown an even deeper anti-patent animus.
Thus, we need an even stronger action this time around.
The only “way out” that I see is to use the constitutionally provided jurisdiction stripping power and remove the non-original jurisdiction of hearing patent appeal cases from the Supreme Court.
Anything less and we will just see that court find another “scrivining” way of reclaiming that power to set power law that is just too addicting for that body to overcome.
step back
August 9, 2015 07:35 amOh Sue Sue Dee You,
There you go again, gettin’ a scientific and all
When as we know that is against the natural flow of Mother Nature’s will.
If Mother Nature had meant for mankind to be rational she woulda given judges fact based brains.
But clearly that ain’t so cause they all make decisions about her “natural” laws as a matter of their own judge made up laws (as a matter of law).
The problem is that you let facts get in your way.
You need to start seeing things in a “new age” light.
We call our new age the “Dark Age”.
Welcome to it and the patent inquisition panels it engenders.
http://patentu.blogspot.com/2015/06/spanish-inquisition-jumps-patentability.html