Hidden away behind the stomach of the typical human torso is a six- to ten-inch long organ known as the pancreas which supports essential bodily functions involved in digestion. The pancreas produces a series of enzymes which break down the proteins, carbohydrates and fats found in our diet to aid in the digestion of those substances. It also plays a pivotal role in regulating blood sugar by producing insulin to lower blood sugar or glucagon to increase blood sugar. Disorders affecting the pancreas include pancreatitis, or an inflammation of the organ, as well as pancreatic cancer, the fourth-most common cause of cancer death among American adults according to the National Pancreas Foundation.
People can also suffer from diabetes when the pancreas cannot generate enough insulin to control blood sugar levels. There are three major types of diabetes: type 1, type 2 and gestational. A 2014 national diabetes report out from the U.S. Centers for Disease Control and Prevention (CDC) found that 29.1 million Americans, or 9.3 percent of the country’s population, have some form of diabetes, and 8.1 million of those cases are undiagnosed. While all types of diabetes can be diagnosed in adults, type 1 diabetes can also affect children and can be a huge concern for parents who are responsible for making sure that their child’s insulin is constantly monitored. According to type 1 diabetes research foundation JDRF, 1.25 million Americans suffer from type 1 diabetes and 40,000 new type 1 diagnoses are made each year.
The discovery of insulin in the early 1920s was a major step in the medical world’s fight against diabetes, which up to that time was a death sentence to patients who had no recourse for regulating blood sugar. Next year, the world could see the next major victory in the battle against diabetes when the first commercially available artificial pancreas systems are available in the United States. Near the end of June, media outlets were reporting that the Irish medical device developer Medtronic (NYSE:MDT) had filed for U.S. Food and Drug Administration (FDA) approval for its MiniMed 670G artificial pancreas system. The device is designed to measure blood sugar levels every five minutes and administer a measured micro-dose of insulin to keep patients within their target range to ward off diabetes symptoms around the clock.
According to the FDA, the ideal artificial pancreas device system will be comprised of a series of devices: a continuous glucose monitoring system, an insulin infusion pump and a blood glucose device which calibrates the monitor. Computer algorithms are also incorporated for accurate blood glucose monitoring and communicating between devices for automated glycemic control.
Continuous glucose monitors have been FDA-approved for about a decade but we’ve only recently reached a point where automated control of insulin is a possibility in the near future according to Aaron Kowalski, chief mission officer and vice president of research at JDRF. “At the beginning, people thought that the monitors weren’t accurate enough to drive the dosing of insulin, and too much insulin at the wrong time can kill,” he said. Today, the use of small body-worn sensors utilizing the same glucose oxidase chemistry as glucose monitoring strips allows monitoring at very short intervals without pricking a finger to draw blood.
Kowalski said that the biggest challenge in developing artificial pancreas systems has been the line between technology and biology. The delivery of insulin through human skin is an non-ideal substitute for insulin produced and delivered by the pancreas, insulin which works more quickly and doesn’t remain in a person’s system. Insulin delivered through the skin, by contrast, takes longer to start working in a person’s body and remains in that body for longer, increasing the chance that a person can feel the negative impacts of too much insulin.
Artificial pancreas systems can provide dramatic improvements to the quality of life of diabetes patients and Kowalski should know, as he himself has had type 1 diabetes for 30 years. “I certainly think that the overnight control is going to be a huge, huge benefit,” he said. “I was fortunate enough to participate in a trial for five days and I was amazed that every single morning my blood sugar was perfect.” Data collected from artificial pancreas device users also shows better outcomes on A1C tests which indicate the risk that a diabetes patient has for developing blindness, kidney failure or other problems associated with diabetes. Kowalski added how his work had put him into contact with diabetes patients who were very frustrated by the variability of the disease despite strict adherence to diet and physical activity.
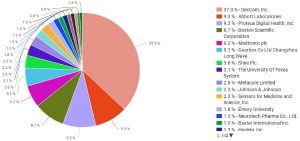 The patent portfolio analysis tools available through Innography show us that there are 503 patents related to artificial pancreas systems which have been issued by the U.S. Patent and Trademark Office. As the pie chart here shows, a full 37 percent of this patent market is held by San Diego, CA-based medical device developer DexCom, Inc. (NASDAQ:DXCM), followed by Abbott Laboratories (NYSE:ABT) of Chicago, IL, and Proteus Digital Health of Redwood City, CA. Medtronic, expected to be the first company to bring an artificial pancreas system to market, sits in fifth place holding 6.2 percent of the artificial pancreas patent market.
The patent portfolio analysis tools available through Innography show us that there are 503 patents related to artificial pancreas systems which have been issued by the U.S. Patent and Trademark Office. As the pie chart here shows, a full 37 percent of this patent market is held by San Diego, CA-based medical device developer DexCom, Inc. (NASDAQ:DXCM), followed by Abbott Laboratories (NYSE:ABT) of Chicago, IL, and Proteus Digital Health of Redwood City, CA. Medtronic, expected to be the first company to bring an artificial pancreas system to market, sits in fifth place holding 6.2 percent of the artificial pancreas patent market.
 A good deal of research into artificial pancreas systems is being conducted at the university level, as is evidenced by U.S. Patent No. 9398869, entitled Method, System and Computer Program Product for Improving the Accuracy of Glucose Sensors Using Insulin Delivery Observation in Diabetes. Issued in late July to the University of Virginia, it claims a processor implemented method for improving the accuracy of a glucose monitoring device by obtaining glucose level readouts and insulin delivery information, performing a series of computations on the glucose and insulin data to estimate the blood glucose of a subject and determining an amount of insulin to be delivered. This system improves blood glucose measurement accuracy by incorporating data on insulin delivery in order to bring the accuracy of continuous glucose monitors more closely in line with test strips which use capillary blood.
A good deal of research into artificial pancreas systems is being conducted at the university level, as is evidenced by U.S. Patent No. 9398869, entitled Method, System and Computer Program Product for Improving the Accuracy of Glucose Sensors Using Insulin Delivery Observation in Diabetes. Issued in late July to the University of Virginia, it claims a processor implemented method for improving the accuracy of a glucose monitoring device by obtaining glucose level readouts and insulin delivery information, performing a series of computations on the glucose and insulin data to estimate the blood glucose of a subject and determining an amount of insulin to be delivered. This system improves blood glucose measurement accuracy by incorporating data on insulin delivery in order to bring the accuracy of continuous glucose monitors more closely in line with test strips which use capillary blood.
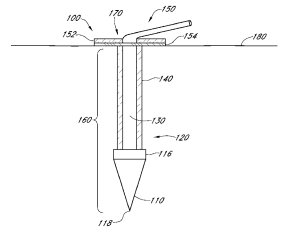 One of the artificial pancreas patents recently issued to DexCom is U.S. Patent No. 9357951, titled Transcutaneous Analyte Sensor. It discloses a sensor device having a sensor unit with a tissue piercing element configured for direct press insertion, an on-skin unit that supports the sensor on an exterior surface of a host’s skin and a skin tensioner that stretches the skin’s surface to create tension sufficient enough to permit needle-less sensor insertion. This innovation seeks to reduce the number of components as well as steps for applying a transcutaneous sensor device to improve the ease of use, especially for new users.
One of the artificial pancreas patents recently issued to DexCom is U.S. Patent No. 9357951, titled Transcutaneous Analyte Sensor. It discloses a sensor device having a sensor unit with a tissue piercing element configured for direct press insertion, an on-skin unit that supports the sensor on an exterior surface of a host’s skin and a skin tensioner that stretches the skin’s surface to create tension sufficient enough to permit needle-less sensor insertion. This innovation seeks to reduce the number of components as well as steps for applying a transcutaneous sensor device to improve the ease of use, especially for new users.
 An improvement to insulin delivery pioneered by Medtronic is at the center of U.S. Patent No. 9364609, entitled Insulin On Board Compensation for a Closed-Loop Insulin Infusion System. This patent protects an electronic insulin infusion device having an insulin reservoir, a processor architecture and a memory element storing processor-executable instructions to perform a method of controlling closed-loop delivery of insulin from the insulin reservoir to a user’s body. The method involves calculating an amount of active insulin in the user’s body, determining an adjusted insulin infusion rate and operating the insulin infusion device to in a closed-loop mode to continuously deliver insulin in accordance with the final determined infusion rate. This device can adjust the amount of insulin based on the activity level of the insulin currently in a person’s body, enabling the use of faster-acting insulin in insulin pumps.
An improvement to insulin delivery pioneered by Medtronic is at the center of U.S. Patent No. 9364609, entitled Insulin On Board Compensation for a Closed-Loop Insulin Infusion System. This patent protects an electronic insulin infusion device having an insulin reservoir, a processor architecture and a memory element storing processor-executable instructions to perform a method of controlling closed-loop delivery of insulin from the insulin reservoir to a user’s body. The method involves calculating an amount of active insulin in the user’s body, determining an adjusted insulin infusion rate and operating the insulin infusion device to in a closed-loop mode to continuously deliver insulin in accordance with the final determined infusion rate. This device can adjust the amount of insulin based on the activity level of the insulin currently in a person’s body, enabling the use of faster-acting insulin in insulin pumps.
The market for artificial pancreas devices including continuous glucose monitors and insulin pumps was $76.5 million in 2015 according to market research firm Grand View Research. In North America, the artificial pancreas device market is expected to grow at a compound annual growth rate of 20.7 percent through 2024 despite stringent FDA regulations. According to Kowalski, these systems may also be capable of restoring pancreatic function to those who have had a pancreas removed, providing greater hope to pancreatic cancer patients.
While Kowalski said that, in theory, artificial pancreas systems could benefit all type 1 diabetes patients, only about 25 percent of diabetes patients in the U.S. are currently meeting recommended glycemic goals for blood glucose levels as a result of their diabetes treatments. “Those who are unwilling to wear the device won’t benefit,” Kowalski said. Still, the advent of artificial pancreas systems should be a great benefit to diabetes patients who are seeking a way to deal with their condition which allows them to enjoy their daily lives to a greater degree.

![[IPWatchdog Logo]](https://ipwatchdog.com/wp-content/themes/IPWatchdog%20-%202023/assets/images/temp/logo-small@2x.png)


![[[Advertisement]]](https://ipwatchdog.com/wp-content/uploads/2023/01/2021-Patent-Practice-on-Demand-1.png)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Patent-Litigation-Masters-2024-sidebar-early-bird-ends-Apr-21-last-chance-700x500-1.jpg)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/WEBINAR-336-x-280-px.png)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/2021-Patent-Practice-on-Demand-recorded-Feb-2021-336-x-280.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/Ad-4-The-Invent-Patent-System™.png)







Join the Discussion
One comment so far.
Jodi
August 27, 2016 08:42 pmWhat I would like to know is would this help with making enzymes. Non diabetic but have to take enzymes because most of the pancreas is gone. This would be fabulous if this would correct this problem.