 Only two infectious diseases have been globally eradicated: smallpox and rinderpest. While both diseases caused people to suffer incredibly different symptoms, the diseases had one element in common. Patents protected the vaccines used to treat smallpox and rinderpest. Intellectual property protection has been crucial for several advances in the medical field, due to the high product development costs and chances of competitors copying the developing technologies. As of 2006, the cost of medical devices in the global marketplace exceeded $260 billion, and has only continued to rise.
Only two infectious diseases have been globally eradicated: smallpox and rinderpest. While both diseases caused people to suffer incredibly different symptoms, the diseases had one element in common. Patents protected the vaccines used to treat smallpox and rinderpest. Intellectual property protection has been crucial for several advances in the medical field, due to the high product development costs and chances of competitors copying the developing technologies. As of 2006, the cost of medical devices in the global marketplace exceeded $260 billion, and has only continued to rise.
However, because the production costs of these medical devices and pharmaceuticals are so high, millions of people around the world are unable to obtain necessary healthcare. For example, as of 2014, close to seventy percent of all cardiac pacemaker sales occurred in the United States and Europe, while several countries in Africa and Asia have absolutely no access to pacemakers. In order to respond to this problem, research scientists have begun developing low-cost medical technologies and using intellectual property rights to give people in developing countries access to adequate healthcare.
Approximately thirty percent of drugs sold worldwide are counterfeits. According to the World Health Organization, the percentage of counterfeit drugs in some developing countries is as high as fifty to seventy percent. Such rampant counterfeiting occurs when “there is [a] lack of intellectual property protection” and regulatory controls preventing the importation or use of substandard medical devices. As a result, strong international intellectual property rights not only allow for innovation in medical technologies, but also deter counterfeiting.
Alerting Patients to Counterfeit Drugs:
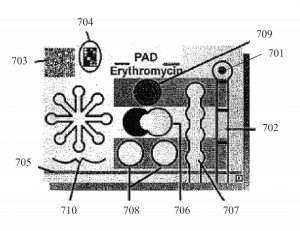
Figure 7 of U.S. Patent No. 9,354,181: an erythromycin PAD that detects counterfeit drugs.
For example, US Patent No. 9,354,181, Analytical Devices for Detection of Low-Quality Pharmaceuticals, was granted in 2016 and discloses a device called PADs. The PADs detect multiple chemicals in a pill or capsule and determine whether the medicines are low-quality or counterfeit goods. They are also the size of an ordinary business card, and give accurate results in five minutes.
As a result, the PADs have been used in developing countries lacking medical technologies and regulatory infrastructure to screen for counterfeit Panadol tablets, along with malaria and flu treatments. Some of the counterfeit drugs PADs screens contain acetaminophen (e.g., Tylenol), which relieves pain and fever, but ultimately does not treat the diseases suffered by the patients. By obtaining patent protection, the researchers “are much closer to getting this technology to market” and having the PADs “be used by pharmacists and others to expose low-quality drugs.”
Spinning Away Malaria Parasites:
The centrifuge, which spins materials in fluids at high speeds, ordinarily costs thousands of dollars and requires electricity. However, Stanford University recently developed an alternative centrifuge that only costs twenty cents and does not require electricity. The Paperfuge is a whirligig made of a disc, such as a button, and a piece of string threaded twice. Ordinary whirligigs can spin from 10,000 to 15,000 RPMs. The Stanford team developed a custom whirligig by fitting a vial of blood and fluids into a paper disc. After pulling on the strings for a few minutes, the Paperfuge achieved RPMs of 125,000 and 30,000 G-forces.
Currently, the Paperfuge is used to separate malaria parasites from blood. Manu Prakash, the developer of the device, owns a registered trademark for Paperfuge. While Prakash and the team currently do not have a patent for the Paperfuge, based on the success of the PADs, patent protection for further advancements in low-cost devices and global health is likely.
Chipping Away at the AIDS virus and Syphilis in Sub-Saharan Africa:
Researchers have also developed low-cost diagnostic tools for treating diseases. In particular, the mChip, developed by Samuel Sia and research scientists at Columbia University, is capable of diagnosing HIV or Syphilis in twenty minutes. The mChip costs $1, and was developed to allow people in regions with poor health facilities to own a portable device and test for HIV/AIDS in their own homes, instead of travelling long distances to health clinics. In particular, developing countries in Sub-Saharan Africa suffer from high rates of HIV/AIDS infection, and do not have easy access to clinics and hospitals for diagnosis or treatment.
When clinical testing was conducted in Rwanda, nearly one hundred percent of the patients were correctly diagnosed when tested for HIV/AIDS. Patients were also accurately diagnosed with syphilis nearly ninety four percent of the time as well.
The success of the mChip in clinical testing led to the team successfully obtaining patent protection. Currently, the mChip has two US patents, US 8,389,272 and US 9,116,148, and has PCT applications pending in China, Canada and Europe. The US patents disclose a method and device for delivering fluids sequentially, which the mChip uses to diagnose HIV/AIDS and syphilis.
Conclusion:
Traditionally, intellectual property rights allowed for scientists and researchers to conduct costly research without having to worry about their competitors copying their work. However, by exclusively focusing on high-cost medical devices and pharmaceuticals, developing countries increasingly lost access to affordable care. If the current trend of developing low-cost technologies for developing countries continues, intellectual property rights will become paramount, in order to eradicate pharmaceutical and medical counterfeiting and improve global health.

![[IPWatchdog Logo]](https://ipwatchdog.com/wp-content/themes/IPWatchdog%20-%202023/assets/images/temp/logo-small@2x.png)


![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/UnitedLex-May-2-2024-sidebar-700x500-1.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Artificial-Intelligence-2024-REPLAY-sidebar-700x500-corrected.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Patent-Litigation-Masters-2024-sidebar-700x500-1.jpg)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/WEBINAR-336-x-280-px.png)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/2021-Patent-Practice-on-Demand-recorded-Feb-2021-336-x-280.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/Ad-4-The-Invent-Patent-System™.png)






Join the Discussion
One comment so far.
Inventor Woes
February 11, 2017 07:22 pm“Such rampant counterfeiting occurs when “there is [a] lack of intellectual property protection””
I’m afraid the rampant counterfeiting is a result of the high price, not lack of IP protection. If anything a strong IP regime would cause even more counterfeiting, especially in the black market.