More than one-quarter of U.S. greenhouse gas emissions in 2014 came from transportation sources according to statistics published online by the U.S. Environmental Protection Agency (EPA). Using a single gallon of gasoline as vehicle fuel results in 8,887 grams of carbon dioxide emissions. Over the course of a year, a typical passenger vehicle emits 4.7 metric tons of carbon dioxide every year according to the EPA.
And yet, carbon emissions from automotive fuel have been reduced drastically over the past few decades in certain respects. Mobile source emissions from cars, trucks and construction equipment have been reduced by 50 percent since 1990 at a rate of 1.5 million tons of hazardous air pollutants (HAPs) per year. Between 1990 and 2005, particulate matter from mobile sources of diesel emissions decreased by about 27 percent. Between 2004 and 2015, fuel economy has increased by 5.5 miles per gallon, an improvement in fuel economy of 28 percent. As of 2015, carbon dioxide emissions from passenger vehicles are at record lows and fuel economy is at record highs, the EPA reports.
Improvements in fuel economy and reduced emissions are largely the result of fuel additives and exhaust catalyst equipment working to clean up the chemicals emitted when fuel is burned. This Friday, April 7th, marks the 25th anniversary of the issue of a seminal patent in the field of automotive exhaust catalyst. The inventor, Indian-born American inventor Haren Gandhi, is an inductee this year into the National Inventors Hall of Fame. Today, we’ll take the opportunity to revisit our Evolution of Technology series to look back at the development of technologies which have left us with cleaner breathing air than we would have had thanks to Americans’ heavy reliance on automobiles.
[JV-1]
The Catalytic Converter Begins the Work of Cleaner Auto Emissions
Although he made massive contributions to the body of knowledge surrounding fossil fuel emissions and techniques for cleaning vehicle exhaust, Gandhi did not make the seminal discovery in the field of cleaner emissions from fuel. That distinction belongs to French-born American engineer Eugene Houdry who invented the catalytic converter, a device still used in vehicles today but first developed to improve the production of gasoline, not clean its exhaust gases. Houdry, an avid auto racing participant, began work while living in France on developing gasoline fuel products which could work better for the high-performance needs of racecars.
In the early 1920s, Houdry learned of a gasoline derived from lignite, a soft, brown form of coal formed from peat which is compressed naturally. Over the following years, Houdry would work with E.A. Prudhomme, the creator of the lignite-derived gasoline, to develop catalytic procedures which improved gasoline production by causing chemical processes to occur in the presence of other chemicals which increase the rate at which those processes take place. By June 1929, Houdry and Prudhomme had developed a method of converting lignite to gasoline which involved using heat to break down the lignite material into oils and tars and then adding a catalyst to the oils which produced hydrocarbons with lower boiling points similar to those hydrocarbons derived from petroleum. This reaction took place inside of what were known as Houdry units, which could produce a few hundred barrels of gasoline per day. Houdry focused on the use of Fuller’s earth as the oil-to-hydrocarbon catalyst.
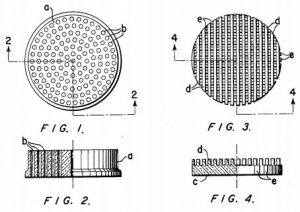 Developments on improved catalytic converters would continue despite the effects of the Great Depression on the global market for oil. In the 1950’s, Houdry, now in America, began to develop catalytic converters not for the production of oil but rather for the elimination of pollutants from automotive exhaust. Around 1950, studies on the effects of smog on human health in Los Angeles led Houdry to work towards creating a catalytic converter unit for automotive exhaust. This April 17th marks the 61st anniversary of the issue of Houdry’s U.S. Patent No. 2742437, entitled Catalytic Structure and Composition. It claims a catalytic structure with a strong support made of substantially impervious material and a catalytically active metal oxide on the external surface of the support to create a unit useful for catalyzing oxidation process for removing heat and pollutants from exhaust fumes.
Developments on improved catalytic converters would continue despite the effects of the Great Depression on the global market for oil. In the 1950’s, Houdry, now in America, began to develop catalytic converters not for the production of oil but rather for the elimination of pollutants from automotive exhaust. Around 1950, studies on the effects of smog on human health in Los Angeles led Houdry to work towards creating a catalytic converter unit for automotive exhaust. This April 17th marks the 61st anniversary of the issue of Houdry’s U.S. Patent No. 2742437, entitled Catalytic Structure and Composition. It claims a catalytic structure with a strong support made of substantially impervious material and a catalytically active metal oxide on the external surface of the support to create a unit useful for catalyzing oxidation process for removing heat and pollutants from exhaust fumes.
Haren Gandhi Reduces Pollution from Exhaust with Three-Way Catalysts
Houdry’s catalytic converter faced challenges in working with automotive exhaust, however, which made the unit somewhat unsuccessful in vehicles. Tetraethyl lead, which had been used to increase the octane of fuels for higher performance settings since the 1920s, fouled the metal oxide used as a catalyst in the converter. It would take the work of Haren Gandhi, another engineer interested in improving the quality of the air we breathe, to turn the automotive catalytic converter into a much stronger tool for reducing pollution.
Born May 1941 in Calcutta, India, Gandhi moved to America during the 1960s after obtaining a degree for studies in chemical technology at the University of Mumbai. In 1967, he began working as a research engineer at the Ford Motor Company, where he would spend the rest of his career racking up honors for his research into automotive exhaust catalyst technology.
Gandhi joined Ford a short time before U.S. government passed the 1970 Clean Air Act into law. This law made a major impact on the automotive industry which had to achieve a 90 percent reduction in both carbon monoxide and hydrocarbon emissions by 1975, as well as a 90 percent reduction of nitric oxide emissions between 1971 and 1976. Gandhi’s work focused on the use of three-way catalysts (TWCs), which were capable of converting the three major pollutants targeted by the Clean Air Act. TWCs can convert carbon monoxide to carbon dioxide, hydrocarbons to carbon dioxide and water, and nitric oxides into nitrogen and water. Much of Gandhi’s work involved the use of precious metals like palladium, a naturally occurring rare metallic element which has low reactivity.
 Gandhi’s pioneering work in this field is reflected in U.S. Patent No. 5102853, titled Three-Way Catalyst for Automotive Emission Control. It claims a TWC for automotive emission control having a mechanical carrier with a support comprised substantially of alumina, a catalytic compound on the support having a major constituent of palladium particles and a discontinuous phase of titanium oxide substantially on or adjacent each exposed particle of catalytic compound. This invention comes after the discovery that the application of small amounts of titania in strategic ways cooperates with palladium to enhance catalytic effects for oxidation and reduction, especially at high temperatures; the use of palladium as the sole catalyst provides a limited catalytic effect in comparison.
Gandhi’s pioneering work in this field is reflected in U.S. Patent No. 5102853, titled Three-Way Catalyst for Automotive Emission Control. It claims a TWC for automotive emission control having a mechanical carrier with a support comprised substantially of alumina, a catalytic compound on the support having a major constituent of palladium particles and a discontinuous phase of titanium oxide substantially on or adjacent each exposed particle of catalytic compound. This invention comes after the discovery that the application of small amounts of titania in strategic ways cooperates with palladium to enhance catalytic effects for oxidation and reduction, especially at high temperatures; the use of palladium as the sole catalyst provides a limited catalytic effect in comparison.
Over the course of his career, Gandhi’s research led to the development of more than 70 technical publications. Along with TWCs, Gandhi developed the concept of oxygen storage in catalytic converters, discovering useful materials which could store oxygen during fuel-lean efforts and then safely release that oxygen later. Gandhi also pioneered conservation and recycling methodologies which provided the catalytic effect required while reducing the need for using precious metals like palladium, platinum or rhodium. He and other Ford researchers also discovered the effects of lead on catalytic materials which served to increase the speed at which lead was banned from gasoline.
According to the National Inventors Hall of Fame, Gandhi earned 61 U.S. patents during his life before he passed away in 2010. In 2002, Gandhi was awarded the National Medal of Technology and Innovation for his contributions to automotive manufacturing, including his work “ensuring the judicious use of precious metals.” During his life, he was appointed to many boards of organizations dedicated to contributing to the field of knowledge in catalytic science and precious metals. In 2008, Gandhi was recognized by the American Institute of Chemical Engineers (AIChE) in its list of 100 Chemical Engineers of the Modern Era.

![[IPWatchdog Logo]](https://ipwatchdog.com/wp-content/themes/IPWatchdog%20-%202023/assets/images/temp/logo-small@2x.png)


![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/UnitedLex-May-2-2024-sidebar-700x500-1.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Artificial-Intelligence-2024-REPLAY-sidebar-700x500-corrected.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Patent-Litigation-Masters-2024-sidebar-700x500-1.jpg)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/WEBINAR-336-x-280-px.png)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/2021-Patent-Practice-on-Demand-recorded-Feb-2021-336-x-280.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/Ad-4-The-Invent-Patent-System™.png)






Join the Discussion
No comments yet.