 While the Patent Trial and Appeal Board (PTAB) has not been friendly to patent owners, to put it mildly, the PTAB has not been inhospitable to pharma patent owners according to a report issued in mid-June by BiologicsHQ, a searchable database of drugs, patents, and companies involved in PTAB inter partes review (IPR) proceedings developed by attorneys at Fitzpatrick, Cella, Harper & Scinto. The BiologicsHQ report shows a much different story in terms of drug patents facing IPR challenges at the PTAB. The report looks at a combination of data sources, including the Orange Book, Center for Drug Evaluation and Research (CDER) listed biologics and statistics on America Invents Act (AIA) trials published by the PTAB. The BiologicsHQ report draws the conclusion that, despite widespread concerns about the PTAB operating as a patent death squad in IPRs, “such concern is not justified for drug patents.”
While the Patent Trial and Appeal Board (PTAB) has not been friendly to patent owners, to put it mildly, the PTAB has not been inhospitable to pharma patent owners according to a report issued in mid-June by BiologicsHQ, a searchable database of drugs, patents, and companies involved in PTAB inter partes review (IPR) proceedings developed by attorneys at Fitzpatrick, Cella, Harper & Scinto. The BiologicsHQ report shows a much different story in terms of drug patents facing IPR challenges at the PTAB. The report looks at a combination of data sources, including the Orange Book, Center for Drug Evaluation and Research (CDER) listed biologics and statistics on America Invents Act (AIA) trials published by the PTAB. The BiologicsHQ report draws the conclusion that, despite widespread concerns about the PTAB operating as a patent death squad in IPRs, “such concern is not justified for drug patents.”
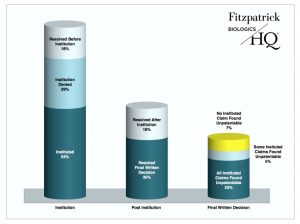 According to the March 2017 IPR statistics issued by the PTAB, 53 percent of IPRs resolved as of March 31, 2017 were instituted; the rest were either denied institution or reached some other resolution prior to the institution decision. 35 percent of all resolved IPRs resulted in final written decisions and 23 percent led to findings of all claims unpatentable. Only 7 percent of all resolved IPRs led to final written decisions finding that no claim was unpatentable, and 5 percent led to mixed claim findings.
According to the March 2017 IPR statistics issued by the PTAB, 53 percent of IPRs resolved as of March 31, 2017 were instituted; the rest were either denied institution or reached some other resolution prior to the institution decision. 35 percent of all resolved IPRs resulted in final written decisions and 23 percent led to findings of all claims unpatentable. Only 7 percent of all resolved IPRs led to final written decisions finding that no claim was unpatentable, and 5 percent led to mixed claim findings.
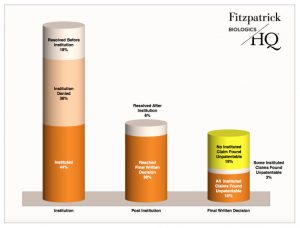 By contrast, drug patents fare better under PTAB scrutiny in terms of having claims upheld. Of the 4,563 resolved IPRs, BiologicsHQ reports that 222 petitions (5 percent) involved patents covering drugs listed in the Orange Book. Focusing on just the IPRs involving Orange Book patents, 44 percent were instituted and 38 percent reached a final written decision, but only 16 percent led to final written decisions where all claims were found unpatentable. No instituted claim was found unpatentable in 50 percent of final written decisions (19 percent of the total number of resolved Orange Book IPRs).
By contrast, drug patents fare better under PTAB scrutiny in terms of having claims upheld. Of the 4,563 resolved IPRs, BiologicsHQ reports that 222 petitions (5 percent) involved patents covering drugs listed in the Orange Book. Focusing on just the IPRs involving Orange Book patents, 44 percent were instituted and 38 percent reached a final written decision, but only 16 percent led to final written decisions where all claims were found unpatentable. No instituted claim was found unpatentable in 50 percent of final written decisions (19 percent of the total number of resolved Orange Book IPRs).
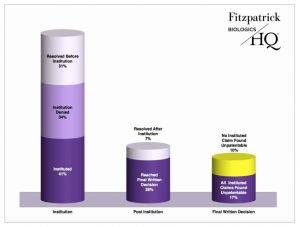 CDER-listed biologic drug patents also survive PTAB challenges better than patents not directed to pharmaceutical drugs, although the number of resolved challenges is quite small by comparison. By March 31, 2017, BiologicsHQ reports that a total of 29 resolved IPR petitions involved patents covering CDER-listed biologics. 41 percent of those petitions were instituted and 28 percent reached final written decisions. 17 percent of all resolved CDER-listed biologic drug IPRs led to final written decisions of all claims unpatentable, and 10 percent led to final written decisions of no claims unpatentable.
CDER-listed biologic drug patents also survive PTAB challenges better than patents not directed to pharmaceutical drugs, although the number of resolved challenges is quite small by comparison. By March 31, 2017, BiologicsHQ reports that a total of 29 resolved IPR petitions involved patents covering CDER-listed biologics. 41 percent of those petitions were instituted and 28 percent reached final written decisions. 17 percent of all resolved CDER-listed biologic drug IPRs led to final written decisions of all claims unpatentable, and 10 percent led to final written decisions of no claims unpatentable.
 In terms of settlements, the BiologicsHQ data indicates that 21 percent of all IPRs involving CDER-listed biologics were settled prior to institution; this accounts for all settlements in IPRs involving CDER-listed biologics. 17 percent of all Orange Book IPRs settled, 12 percent prior to institution and 5 percent after. For all technologies, 30 percent of all resolved IPRs reached settlements, but only 17 percent were settled prior to institution.
In terms of settlements, the BiologicsHQ data indicates that 21 percent of all IPRs involving CDER-listed biologics were settled prior to institution; this accounts for all settlements in IPRs involving CDER-listed biologics. 17 percent of all Orange Book IPRs settled, 12 percent prior to institution and 5 percent after. For all technologies, 30 percent of all resolved IPRs reached settlements, but only 17 percent were settled prior to institution.
When comparing final written decisions among all IPRs, IPRs relating to Orange Book patents, and IPRs relating to CDER-listed biologic drug patents, Orange Book and CDER-listed biologic drug patents are more likely to escape with all claims intact. For all resolved IPRs, 23 percent led to final written decisions of all claims unpatentable. That’s a higher percentage than the 16 percent of Orange Book IPRs, and 17 percent of CDER-listed biologic drug IPRs that led to final written decisions where all claims are unpatentable. The 7 percent of all IPRs that led to final written decisions of no claims unpatentable is less than the 10 percent of CDER-listed biologic drug IPRs, and the 19 percent of Orange Book IPRs, that met the same fate.
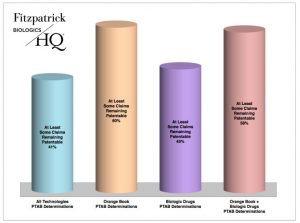 Finally, the BiologicsHQ report looks at the result of determinations made by the PTAB alone, weeding out settlements or other terminations resulting as a decision between the two parties. When looking at all technologies, 41 percent of the IPRs terminated as a result of a PTAB determination resulted in at least some claims remaining patentable. This includes institution denials as well as final written decisions of no claims unpatentable or mixed claim findings. Overall, 58 percent of IPRs involving drug patents resolved by the PTAB resulted in some claims remaining patentable: 60 percent for Orange Book IPRs and 45 percent for CDER-listed biologic IPRs.
Finally, the BiologicsHQ report looks at the result of determinations made by the PTAB alone, weeding out settlements or other terminations resulting as a decision between the two parties. When looking at all technologies, 41 percent of the IPRs terminated as a result of a PTAB determination resulted in at least some claims remaining patentable. This includes institution denials as well as final written decisions of no claims unpatentable or mixed claim findings. Overall, 58 percent of IPRs involving drug patents resolved by the PTAB resulted in some claims remaining patentable: 60 percent for Orange Book IPRs and 45 percent for CDER-listed biologic IPRs.
There could be multiple factors in play as regards to why drug patents seem to fare better at the PTAB compared to all other technologies according to Robert Schwartz, partner at Fitzpatrick. He indicated that patent claims covering pharmaceuticals might be more carefully drafted or there may be more opportunities for patent owners to demonstrate nonobviousness through evidence of superior unexpected results. “It has a lot to do with the fact that pharmaceutical technologies in general are more complicated than other technologies,” Schwartz said.
Schwartz said that a lower rate of trial institution for Orange Book and CDER-listed biologic patent IPRs might be a contributing factor as to why more of those patents survive with at least some claims intact. “The name of the game is trying not to get instituted,” Schwartz said. “Once you’re instituted, your chance of having claims survive gets worse.” That wasn’t to say that a decision to institute necessarily leads to claims being invalidated, he added. “In many cases, they’ll institute a trial if evidence on both sides makes it difficult to have a clear winner. The PTAB will give each side an opportunity to flesh out their arguments.”
Given that statistics on institution decisions published by the PTAB often paint a misleading picture as to the number of challenged claims which are eventually invalidated, it’s possible that a larger percentage of Orange Book and CDER-listed biologic patent claims are being invalidated than the BiologicsHQ report suggests, because each petition is counted separately even if there are multiple challenges to the same patent with different outcomes. April Breyer, an associate at Fitzpatrick and one of the authors of the BiologicsHQ report, noted that the reason the BiologicsHQ team based their data on the PTAB statistics is that they wanted to do an apples-to-apples comparison with what was being reported by the agency.
Breyer noted that a lot of legwork was involved in compiling this data including reading each petition to find Orange Book patents and identify petitions related to biologics. “Nobody has taken the USPTO statistics for all technologies and broken out petitions like we have specifically for drug patents,” Corinne Atton, an associate at Fitzpatrick and the co-author of the report said. In addition to the recent report, BiologicsHQ also provides a searchable online database regarding CDER-listed biologic drug patent challenges at the PTAB. As trends appear, a statistics page on BiologicsHQ collates and reports on those trends, providing metrics that may be useful for making strategic determinations.

![[IPWatchdog Logo]](https://ipwatchdog.com/wp-content/themes/IPWatchdog%20-%202023/assets/images/temp/logo-small@2x.png)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Patent-Litigation-Masters-2024-sidebar-early-bird-ends-Apr-21-last-chance-700x500-1.jpg)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/WEBINAR-336-x-280-px.png)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/2021-Patent-Practice-on-Demand-recorded-Feb-2021-336-x-280.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/Ad-4-The-Invent-Patent-System™.png)






Join the Discussion
36 comments so far.
Tesia Thomas
July 18, 2017 07:56 pmThe “predictability” argument also further relates chemistry to weather and thus further to being “natural phenomena.”
A chaotic system, nonlinear dynamics, complexity.
Weather is nothing but chemistry, too.
Definitely a 101 rejection.
Tesia Thomas
July 18, 2017 07:38 pmYeah @Anon. I totally agree that chemists are given too much leeway.
If you got a degree in this field and you’re saying it’s too unpredictable then what the he** have you learned?
You can’t learn the unpredictable.
Mastery of the field means doing more than guess and check and then claiming unpredictability.
There are many laws in chemistry, all depending on conditions. Boyles law, Avogadro, ideal gas law, etc.
It’s just not sufficient.
If unpredictability is really the nature of chemistry then there is no “one skilled in the art.”
Everyone’s clueless.
Tesia Thomas
July 18, 2017 07:29 pmChemistry is only regarded as unpredictable because humans haven’t tested the limits.
Chemicals depend heavily on their environment.
Algorithms can be equally unpredictable, called pseudorandom and the basis for cipher/crypto.
Anyway, no one knows why anything works or happens until they try it.
That is the reason for experimenting and trial and error.
Every inventor undergoes this.
Once the Bunch o Ballons inventor found a way to fill multiple balloons at once, it was obvious.
Once penicillin was found out, it’s obvious.
Unpredictablility is the nature of all inventions.
If it was really predictable, you would’ve already had it.
1+1=3 in biology (reproduction)
1+1=2 (math)
1+1=1 (consuming something)
AI being unpredictable due to algorithms:
http://www.businessinsider.com/google-just-proved-how-unpredictable-artificial-intelligence-can-be-2016-3
Stretching human imagination and thinking outside the box is unpredictable.
The only reason math appears to be predictable is that it is carefully defined.
I just think pharma should be treated the same.
Predictability means constraining variables. The problem with chem is that no one has done that.
People just combine ingredients and hope they get intended or new results.
Chemistry is a lot of ‘solution in search of a problem.’
Chemists mix tons of different mixtures, test them and then add more stuff to them, mix, test…
That’s why it’s called “drug DISCOVERY.”
It’s total guess and check most often.
But, again, as Thomas Edison(or whoever) said,
“I didn’t fail 100 times, I found 99 ways that didn’t work. And one that did.”
Or something like that.
And he was talking about mechanical tech.
Everything new is unpredictable.
Chemists just don’t try to understand enough in my opinion.
The reason math has so many rules is people really put thought into understanding the relationships between numbers.
If you don’t know why combining X and Y worked then why did you combine it?
Not enough rules in chemistry doesn’t mean unpredictability. It means laziness or uncharted territory of understanding.
Anon
July 18, 2017 06:26 pm“and if this skilled artisan lives in the same world of uncertainty, then you’re good to go. ”
We will have to agree to disagree on this point, explicitly when it comes to the possession requirement.
As I indicated (and recognizing that the term may have a slightly different meaning as a term of art), the term “unpredictability” should probably NOT be used because doing so confuses “ease” with any sense of required “exactness.”
Yes, I “get” that “exactness” may vary from art field to art field, but as the term “unpredictable” denotes (and again, perhaps overreading that term from a non-term of art angle), one is not granted patents out of not knowing (even product by process patents have to have a KNOWN – and repeatable – process), and “ease” and getting actual factual matters wrong should never be tolerated.
And frankly, the comment of “it’s a tough standard to meet when mother nature makes the calls” does not cut it – this TOO leads to the view that chem arts are being given a mulligan that NO OTHER art fields get – and that is plainly NOT how the patent laws are drawn up.
***
Beyond that quibble, the other views being exchanged here should be looked at with merit: i.e., the points that even I and Tesia are finding common ground upon.
CP in DC
July 18, 2017 05:57 pmAnon
Well, this spiralled out of control more than I expected.
The original argument addressed why pharma and biotech patents did better at the IPR stage. IPR is instituted based on anticipation or obviousness. Anticipation in pharma is addressed in prosecution, easy to find it. Obviousness is very subjective, what is “easy to combine” or “obvious to try?” The conversation addressed why merely combining individuals elements rarely invalidates a patent. Obviousness is defeated by unpredictability in pharma.
When I speak of unpredictability, I mean that the science is unable to accurately predict what will happen based on prior knowledge. Extrapolation based upon current knowledge does not always work. Yeah, it can predict the simple but not much after that. That’s why pharma patents have many examples. Also, once you add the human body to the equation, the variables spiral out of control and what worked well in a test tube or cellular level, fails. Basically, you have to try it to figure it out.
If you want exactness (like math) chemistry can’t provide it. Chemists accept that not all reactions yield 100% (conservation of matter be damn, a lot of it are impurities or junk you can’t figure out). Chemists accept that writing a formula encompasses isomers if available. It’s too time consuming to identify everything and anything and sometimes impossible. Chiral compounds, like thalidomide, can exist pure one isomer, pure the other isomer, a 50:50 mixture, or 80:20 mixture or other mixtures. Even repeating the same reaction can give you different yields and mixtures and you don’t know why. So we live in that world of uncertainty. Unless there is a different biological effect, one continues with the work.
I mentioned the analytical techniques because you can’t tell what you have unless you can see it. Due to complexity and expense, sometimes only a few techniques are used. Even light rotation in unpredictable, you don’t know which isomer rotates light leftward or rightward until you run the test. Impure mixtures rotate light less in one direction than a pure isomer.
Patent law does not require you to know how your compounds work or even what they are. Product by process claims arose because people couldn’t accurately describe what they had. 112, description is an issue, but it doesn’t require the exactness found in scientific literature, just enough so the skilled artisan understands and if this skilled artisan lives in the same world of uncertainty, then you’re good to go.
I didn’t mind the comment, I understood where it was coming from. I know exactness is desired, but it’s a tough standard to meet when mother nature makes the calls.
Invention Rights
July 18, 2017 04:39 pmThese invalidation rates are nothing to celebrate. It’s like Toyota “only” recalled 7 million airbags compared to Honda’s 10 million. Recalling patents after they are issued is devastating to the patent system. Even a 1% defect rate is intolerable.
Anon
July 18, 2017 04:22 pmmike,
Your comment of “that chemistry has relied for decades on a system of nomenclature in which a name unambiguously describes a physical structure” simply falls short – leastwise when a path of “ease” is being taken.
😉
Anon
July 18, 2017 04:21 pmCP in DC @ 26,
Thanks again for the insights. But does this not prove my point? You say “for ease” when that “ease” is simply not factually correct.
Also, I think it matters not at all that there are scientific methods (the “many analytical techniques”) that cannot tell a “right hand” from a “left hand” when the fact remains that a true, complete and absolute description of what you have clearly shows that the items are just not “the same.”
“These isomers arise from the three dimensional arrangement of the molecule.”
Yes – but that does not change the fact that they are indeed not the same, now does it? Sure, taking “easy” shorthand “common vernacular” may make it seem that they are the same, but such “they are the same” when they are NOT the same when it comes to legal documents (of which patents are) really is not good enough (if for no other reason than 35 USC 112).
***
Also, the shorthand of “unpredictability” is a bit offensive when what is really being discussed is simply a lack of necessary (albeit not always) discipline to accurately describe what some call “objective physical structure.”
It is far less “unpredictable” and far more simply not being clear what is the actual structure being focused upon. Calling it “unpredictable” is more than just a little bit of a mulligan that is not provided other art units. If you really mean something that requires a three (or perhaps even four) dimensional description, then “ease” is no excuse for not providing what is required.
Whenever I hear the word “unpredictable,” I hear an excuse of “we really don’t know what is going on.”
You do not possess something when you “really don’t know what is going on,” now do you?
(this is NOT meant as a harsh criticism of you personally, by the way)
mike
July 18, 2017 04:18 pmI think there are some unique factors that make many drug patents comparatively robust. The first is that chemistry has relied for decades on a system of nomenclature in which a name unambiguously describes a physical structure. When you claim a novel compound, or a group of novel compounds, 90% of your description writes itself. Not only is it difficult to write a claim for a pure compound that would draw a written description rejection, but the mass of reference material based on that system of nomenclature makes it easy for both patentee and examiner to conduct rapid and near-conclusive searches to determine whether the compound is novel. Any challenge based on anticipation is also nearly always glaringly obvious – either the prior art describes the compound, or it doesn’t.
Second, the USPTO has long accepted the unpredictability of the chemical arts, both in structure/activity relationships as alluded to by CP in DC, and in synthesis. This reduces the number of absurd analogies and hindsight reasoning popular in many other classes for formulating obviousness rejections. An undue level of experimentation is almost a given for even apparently minor changes in structure.
The result is that drug patents for the actual drug compounds tend to be very narrow, claiming a finite (although sometimes very large) number of very precisely defined embodiments. With challenges based on written description or anticipation so easy to judge and those based on obviousness so difficult to assert, it isn’t hard to see why these would be comparatively strong patents.
Anon
July 18, 2017 03:36 pmSVI @ 17,
Per my comment at 19, chirality is an indication that one isomer is NOT the same as another (the TOTAL molecule – when fully and accurately described; their absolute description – are different, and it is only the “shorthand” version of describing a more generalized “class” that leads to a confused statement of “the molecules are the same.”
From the wiki on chirality: Chiral molecules and ions are described by various ways of designating their absolute configuration, which codify either the entity’s geometry or its ability to rotate plane-polarized light, a common technique in studying chirality.
CP in DC
July 18, 2017 03:36 pmAnon
It’s mostly for simplicity, ease, and convenience that chemists use a single compound to represent all isomers.
For chemists, we call both enantiomers the same molecule if it has the same parts, attached in the same order, and in the same way. Many analytical techniques cannot differentiate between the two: nuclear magnetic resonance, high pressure liquid chromatography (unless you use a chiral column that works), most chromatography, melting point, and there are probably more. X-ray diffraction does distinguish, but it’s hard to make everything into a crystalline form, some compounds just don’t crystallize. It’s a trial and error process.
These isomers arise from the three dimensional arrangement of the molecule. Sometimes this has biological effects and sometimes it does not. So more often than not, people just use the mixture and present it as one compound. It’s just easier and generally chemical compounds are written as two dimensional figures.
Should there be a difference and many look at this issue (especially after thalidomide) then it is addressed. Otherwise it is ignored for simplicity. Some molecules have 4 or 5 chiral centers. Four centers are 16 different three dimensional structures and 5 is 32, and so on. So for convenience, we don’t write out all 16 or 32 variations when speaking of the “compound.”
Thalidomide was used as an example of the unpredictability of drugs. And this unpredictability is the savior and destroyer of pharma patents.
Tesia Thomas
July 18, 2017 03:34 pmI agree. All we have is what nature gives us. Everything comes from something around us. And we combine everything differently to make new tools.
Just like melting silver eating utensils to make silver bullets, as an example. It’s all matter transformed.
But then we’re anti entire patent system.
Welp! It’s all matter. Nothing new here!
Anon
July 18, 2017 03:31 pm“if her” ==> “if he”
Anon
July 18, 2017 03:28 pmTesia,
There may be a point of view that we share perhaps some common ground.
Namely, “Elements and chemicals are building blocks of pharma and materials.”
You may be interested in a “logic gone bad” that has been coined as: The Big Box of Protons, Neutrons, and Electrons.
As you may not know, Mr. Quinn is a bit of Nerd (meant in a complimentary manner), although I cannot remember if her prefers Star Trek over Star Wars (or the opposite). Regardless, consider the Star Trek replicator. There, you have a machine that outputs any combination of protons, neutrons and electrons. All that you need is a replicator and a “big box of protons, neutrons and electrons” and what you “have” is any (and all) configurations possible from those three (classical physics) basic building blocks.
Much as M A N Y anti-software patents use “logic gone bad,” if that logic is followed to its ends, then ALL matter falls out of being patent eligible.
Of course, such result is absurd, and leads to the conclusion that the “logic gone bad” is what is absurd. But that still will not stop those who are anti-software patent from attempting to use “logic gone bad.”
Tesia Thomas
July 18, 2017 03:22 pmIsomers are the equivalent of…
1-4n and 4n-1
Both are expressions comprising the same parts but in different arrangement.
As you can see that makes all the difference in what you get as a result.
I agree with you @19 too Anon! 🙂
Tesia Thomas
July 18, 2017 03:19 pmLol Anon I knew you’d say that!
*eye roll*
At least we agree on this! (@10)
Anon
July 18, 2017 03:10 pmTesia,
I am amused that you want to give math lessons.
Truly, I laughed out loud.
🙂
Anon
July 18, 2017 03:09 pmCP in DC,
Many thanks – I was not aware that in the thalidomide situation that there were more than one “item” and that one “item” was good while the other was bad.
The wiki reference was also helpful and contains (footnotes omitted, emphasis added):
Pharmacology
The precise mechanism of action for thalidomide is unknown although efforts to identify thalidomide’s teratogenic action generated 2000 research papers and the proposal of 15 or 16 plausible mechanisms by 2000., As of 2015 the main theories were inhibition of the process of angiogenesis, its inhibition of cereblon, a ubiquitin ligase, and its ability to generate reactive oxygen species which in turn kill cells.
Thalidomide also binds to and acts as an antagonist of the androgen receptor (AR) and hence is a nonsteroidal antiandrogen (NSAA) of some capacity. In accordance, it can produce gynecomastia and sexual dysfunction as side effects in men.
Thalidomide is provided as a racemic mixture of two enantiomers; while there are reports that only one of the enantiomers may cause birth defects, the body converts each enantiomer into the other through mechanisms that are not well understood.
Chemistry
Thalidomide is racemic; while the S-thalidomide is the bioactive form of the molecule, the individual enantiomers can racemize to each other due to the acidic hydrogen at the chiral centre, which is the carbon of the glutarimide ring bonded to the phthalimide substituent. The racemization process can occur in vivo.
***
Separately though, do you really have “the same molecule” (your comment of “Yet another unexpected property all in the same molecule.“)…?
To wit, both a left hand and a right hand are both considered “hands,” but as you aptly pointed out, a left hand is not the same as a right hand.
Would it not be more correct to say that the description of “the same molecule” is not accurate enough to reflect actual molecular differences?
In other words, it may be a “common vernacular” to say “different individual enantiomers are the same molecule,” but is not the physical truth the fact that the molecules are NOT the same? Much like a right hand is NOT the same as a left hand?
Tesia Thomas
July 18, 2017 02:54 pm@SV Inventor
I don’t see it. It’s all steps and processes. Chemistries and algorithms are recipes.
You can change the chemistry of something by putting it a flask of a different material. For instance, polymer synthesis in metal vs glass flask because glass is inert. And if metal is used some of those metal elements can get into your product and completely change the product. I’ve worked in both fields- chem and comp sci.
Math is building blocks of algorithms.
Elements and chemicals are building blocks of pharma and materials.
An algorithm that uses the expression
1-4n to solve a problem
is different from one that uses 4n-1
One might be faster or slower, bettet or worse.
Just as isomers beget different chemistries so do different arrangements in math.
I think we should make SCOTUS and the House look at the facts.
Pharma is no different than software.
Element- compound- drug/material
Number- equation- algorithm/software
Pharma needs more 101 objections. Lol
Silicon Valley Inventor
July 18, 2017 02:42 pm@Tesia #15, no I believe that achieving safety and efficacy by accommodating chirality in thalidomide is more about manufacturing steps and processes. It is not at all the same as software processes because you set up software to achieve a result that does not compromise much of your “yield”. But patenting the steps are indeed analogous and it seems there is no legal reason why pharma patents should be treated any differently by the courts.
Tesia Thomas
July 18, 2017 02:21 pmOr even PEDMAS.
Put parenthesis somewhere. Change an entire equation thus algorithm.
Tesia Thomas
July 18, 2017 02:13 pmAs far as isomers, those can be related to algebra.
3x + 4 = 10
10 = 4 + 3×
Or assigning variables vs comparing them
a=b
a==b
Or even a simple negative positive thing
1-4
Is a different expression than
4-1
Only due to arrangement
Tesia Thomas
July 18, 2017 01:21 pmReally algorithm=chemistry
All are a chain or compound of tasks that must execute just so in order to combine to obtain a specific result.
CP in DC
July 18, 2017 01:20 pmAnon
The thalidomide cases were one example where two isomers of the same compound produced two very different results. In this case, isomers are structures have the same items arranged differently. Let me explain, look at your hands, four fingers, one thumb, a palm, and the back of your had. Now place your hands in front of each other (palms facing each other), they are mirror images. Now place your hands over each other with your palms facing the same direction…. what do you find? Your thumbs are on opposite sides.
Some chemical compounds have this property. Like your hands, some chemical compounds function the same, other do not.
Thalidomide was such a compound. It was given to women for morning sickness. One isomer worked wonders, the other was toxic. The following is from wikepedia:
Shortly after the drug was sold in West Germany, between 5,000 and 7,000 infants were born with phocomelia (malformation of the limbs). Only 40% of these children survived.[4] Throughout the world, about 10,000 cases were reported of infants with phocomelia due to thalidomide; only 50% of the 10,000 survived.
So even though compounds look the same and have the same parts, they can act differently, very differently. Now, imagine what happens when you start to change parts, properties can change. You do not know until you run a test.
Thalidomide is now used to treat cancer with a black box warning. It means, if your must, you can use it, but not if you are thinking of having children. Yet another unexpected property all in the same molecule.
This unpredictabilty is what makes pharma patents difficult to write, prosecute, and get broad coverage.
Tesia Thomas
July 18, 2017 01:19 pmTo clarify my point, @CP in DC,
All elements are naturally occurring, right?
All chemistry is building on or integrating a bunch of elements, right?
Seems like an Alice objection could apply to that too.
Everything comes from something and chemistry iterates to minutia -polydispersity, catalyst, etc.
An algorithm can be efficient and effective or not depending on its structure. Some algorithms are really slow and use a lot of processing power and will make your computer crash. Some are fast and low system drain.
All depending on a simple equation or line of code- minutia.
Can’t we liken that to drugs being effective, having bad symptoms ( computer crashing), or being slow to action. Some of the common defenses of new pharma…
All depending on a simple chain reaction here or catalyst there- minutia.
Anon
July 18, 2017 01:01 pmCP in DC,
For personal interests, can you flesh out your comment of “Minor variations can lead to disaster, see the thalidomide cases.“…?
Thanks
Anon
July 18, 2017 12:58 pmThree words:
Divide
And
Conquer
Tesia Thomas
July 18, 2017 12:47 pmI can see that.
But in chemistry, people will patent polydispersity, claim a new way of polymerization, different catalyst, etc
Minute details to work around the IP.
Drugs do this too. Patents for taking pills vs liquids. Layers with time release.
I can see a difference with mechanical tech. But even then some things have to be extremely precise. Software, algorithms is just as precise.
CP in DC
July 18, 2017 12:36 pmI work with pharma and biotech patents and I know the work required to file, prosecute, and get the claims allowed. Unlike other technologies, the unpredictability of drugs requires more experimental data; data that is difficult to write about, explain, and present in an overly broad manner without running into problems. Too broad and you get an obviousness rejection, too narrow and you invite work-arounds. Just look at the Millenium Pharm v Sandoz case that came out yesterday to see how courts think about pharma claims.
What is complicated is to accurately predict how the technology will work. Minor variations can lead to disaster, see the thalidomide cases. Naturally, the PTO (and other foreign jurisdictions) requires more proof of concept and generally allows narrow claims. Yes secondary considerations are huge in this field.
As a general proposition, a drug is protected by few patents compared to a phone, TV, etc. It’s difficult to generate the necessary data for new filings. So patent applications are drafted with this in mind. Should the drug be profitable, the generics enter the market by challenging validity. See Hatch Waxman litigation to get an idea. Therefore, applications are drafted to protect the products knowing a challenge is very likely. It is only recent that generics use IPR, and they have had better success than in district court for the reasons we all know.
Patent eligibility is a problem, the earlier PTO guidelines were a joke. But word was that drugs were too expensive and there was a need to stop patent protection, but this is a conversation for another day.
Tesia Thomas
July 18, 2017 11:29 amYeah @angry dude & @Curious I was asking because I didn’t see it. Haha
Curious
July 18, 2017 11:09 amHe indicated that patent claims covering pharmaceuticals might be more carefully drafted or there may be more opportunities for patent owners to demonstrate nonobviousness through evidence of superior unexpected results. “It has a lot to do with the fact that pharmaceutical technologies in general are more complicated than other technologies,” Schwartz said.
From the outside looking in, patents claims covering pharmaceuticals appear to be quite simplistic. The elements are fairly well defined. Regardless, I’m not overly impressed with the level of claim drafting. The evidence of superior unexpected results, however, is a huge difference. However, to say that pharmaceutical technologies are more complicated than other technologies is a bunch of hogwash. There may be some simplistic technologies out there that the average person could understand. However, I’m entirely sure that every field has certain technologies that only those extremely familiar with that field would understand.
If you want to know what the PTAB treats pharma better, you have to understand why the PTAB treats other technologies differently. The technology with the highest monetary value is computer related, which is why a lion’s share of IPRs are in that field. For the most part, you aren’t going to IPR a patent on little used technology with little economic value. With that in mind, Michelle Lee game to the USPTO (via the White House) from Google with an implicit mandate to make computer-related inventions about as worthless as possible. However, Google (and hence Michelle Lee) really didn’t care to much about pharma. As such, I’m sure Michelle Lee was pretty much hands off when it comes to that technology.
If, instead of Michelle Lee, you had someone at the reigns of the USPTO who was former executive at a generic pharma company, I would wage good money that the percentage of pharma patents lost at IPRs would be MUCH HIGHER than they are today.
angry dude
July 18, 2017 11:03 amTesia Thomas@3
“…pharmaceutical technologies in general are more complicated than other technologies,” Schwartz said.”
Except for they are not…
He is full of sh1t. that’s all to it.
Don’t believe a single word coming from those liars
shame, shame, shame…
Tesia Thomas
July 18, 2017 10:22 amHe indicated that patent claims covering pharmaceuticals might be more carefully drafted or there may be more opportunities for patent owners to demonstrate nonobviousness through evidence of superior unexpected results. “It has a lot to do with the fact that pharmaceutical technologies in general are more complicated than other technologies,” Schwartz said.
So complicated to synthesize so a lot of steps in the method or ingredients in the apparatus?
What about the simple combination drugs?
angry dude
July 18, 2017 09:53 amNo kidding…
Without enforceable patents US pharma industry will collapse in a matter of weeks, buried under mountains of cheap imported generics
Same with biotech
For the rest of us, poor tech animals, trade secrets forever.
shame, shame, shame…
Valuationguy
July 18, 2017 08:39 amAhh….realpolitiks in action.
The PTAB administrative judges (being political animals….i.e. appointed with the blessings of their political patrons) knows that drug patents are THE hot button which can trigger a significant part of Congress to turn away from its ‘kill patents’ (or efficient infringement as it is called)….resulting in reform which would curb the PTO’s power. Thus they get the soft-glove treatment compared to other industries.
Bemused
July 18, 2017 08:34 amThese results might also be due to the fact that Kyle Bass was filing IPRs against pharma drugs in order to short their stock. But that would mean that the PTAB/PTO was skewing results in favor of pharma companies as a result of political pressure and we know the PTAB/PTO wouldn’t do that, right?