“Particularly in the area of diagnostics in the life sciences, where Cleveland Clinic patents have been challenged and overturned in court, the uncertainty of patent protection makes it less likely we and other inventors will make the investments to make new advances commercially available.” – Peter O’Neill, Executive Director, Cleveland Clinic Innovations
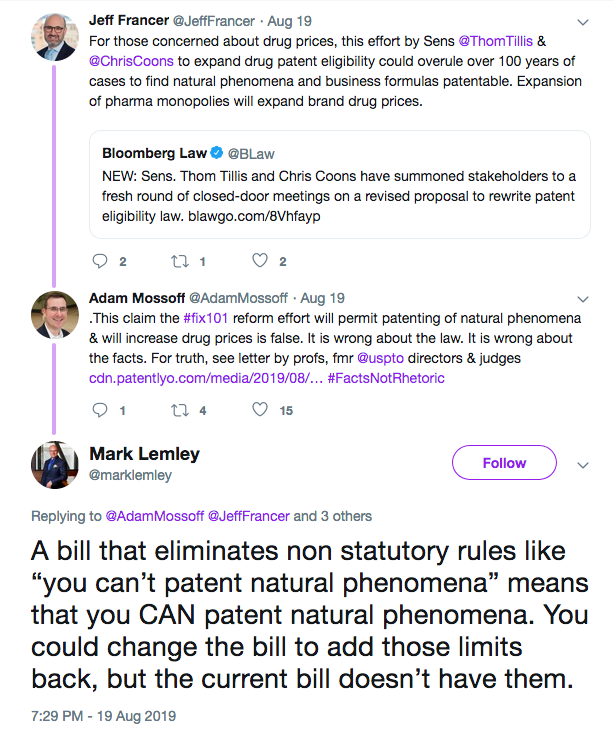 Last month, we reported on the responses submitted to Senator Richard Blumenthal (D-CT) by panelists who participated in the June hearings on the state of U.S. patent eligibility, held by the Senate Judiciary Committee’s Subcommittee on Intellectual Property. Along with Senators Thom Tillis (R-NC) and Mazie Hirono (D-HI), Senator Blumenthal entered a series of questions for the record to be answered by certain participants. While movement on the bill appears to be stalled for the time being, with reports that Tillis and Senator Chris Coons (D-DE) have reinstituted the stakeholder roundtables that led up to the draft bill and hearings in the first place (see related Twitter debate here and pictured), it’s worth reviewing some of the responses to Tillis’ questions as the IP community waits for the next move. From David O. Taylor’s statistic that 62% of investors he surveyed said they were less likely to invest in companies where patent protection is not available, to Bob Armitage’s characterization of the draft bill’s revision to Section 112(f) as “perfect,” to the Cleveland Clinic’s statement that they are currently less likely to make the necessary investments to bring new advances in diagnostics to market, these responses are a reminder of what’s at stake.
Last month, we reported on the responses submitted to Senator Richard Blumenthal (D-CT) by panelists who participated in the June hearings on the state of U.S. patent eligibility, held by the Senate Judiciary Committee’s Subcommittee on Intellectual Property. Along with Senators Thom Tillis (R-NC) and Mazie Hirono (D-HI), Senator Blumenthal entered a series of questions for the record to be answered by certain participants. While movement on the bill appears to be stalled for the time being, with reports that Tillis and Senator Chris Coons (D-DE) have reinstituted the stakeholder roundtables that led up to the draft bill and hearings in the first place (see related Twitter debate here and pictured), it’s worth reviewing some of the responses to Tillis’ questions as the IP community waits for the next move. From David O. Taylor’s statistic that 62% of investors he surveyed said they were less likely to invest in companies where patent protection is not available, to Bob Armitage’s characterization of the draft bill’s revision to Section 112(f) as “perfect,” to the Cleveland Clinic’s statement that they are currently less likely to make the necessary investments to bring new advances in diagnostics to market, these responses are a reminder of what’s at stake.
Questions from Chairman Tillis

David Taylor
David O. Taylor, Co-Director of the Tsai Center for Law, Science and Innovation and Associate Professor of Law, Southern Methodist University Dedman School of Law
Professor Taylor, you’ve also written extensively on the subject of patent eligibility and the current confusion created by the judicially created exceptions… What did you mean by patent law is in a state of crisis?
“First, there is significant confusion… Second, the confusing test lacks administrability… Third, the test generates incorrect results, particularly in the life sciences… Fourth, all this confusion and ineligibility has negatively impacted investor behavior… Beyond these significant problems, there is also a crisis because the Supreme Court is highly unlikely to correct these problems.”
What risks of under investment in research and development has your research demonstrated? Can you give some examples for the Committee of some of the major areas of innovation that are at risk?
“I conducted a survey of 475 venture capital and private equity investors to study the impact of the Supreme Court’s patent eligibility cases on investment firms’ decisions to invest in companies developing technology…
“Overall 74% of the investors agreed that patent eligibility is an important consideration in firm decisions whether to invest in companies developing technology; only 14% disagreed… [O]verall 62% of the investors agreed that their firms were less likely to invest in a company developing technology if patent eligibility makes patents unavailable, while only 20% disagreed…
“[I]nvestors… overwhelmingly indicated that the elimination of patents would either somewhat decrease or strongly decrease their firm’s investments in the biotechnology (77%), medical device (79%), and pharmaceutical industries (73%)…
“[I]t is important to highlight that the results show the Court’s decisions have negatively impacted each and every area of technological development studied. And, as a consequence, the results do support the idea that the time has come for Congress to at least consider overturning the Supreme Court’s new eligibility standard to prevent additional lost investment in technological development in the United States.”

Bob Armitage
Robert Armitage, Consultant, IP Strategy and Policy
From the beginning of our roundtable process you were advocating making tweaks to Section 112(f) as an alternative way to address the concerns some have regarding the issuance of overbroad software patents. As you know, our proposal attempts to do just that. Can you explain to us very briefly your thought process on why a 112(f) fix is a necessary and appropriate compliment to any 101 reform?
“Claims of such potentially stunning breadth have been sought—and secured—in all areas of technology, including the biopharma arena. Eli Lilly and Company, as an example, was sued under U.S. patent 6,410,516 with claims directed to ‘NF-?B induction’ processes…
“It took eight years from the day the patent issued before the patent was finally invalidated as having only insufficiently disclosed claims under the ‘written description’ requirement in 35 U.S.C. § 112(a)…
“I have no doubt whatsoever that the U.S. patent system would have better served both Ariad and Lilly had amendment to 35 U.S.C. § 112(f) proposed to accompany the § 101 reforms now before this Subcommittee been part of the patent law in 2002. Ariad would have secured a valid patent with claims extending to the specific means it disclosed in its patent and any equivalents thereto. Lilly would have been spared years of litigation and its multi-million dollar defense efforts. This is what I call a win-win-win outcome.”
What do you think about our proposed 112(f) language? Do you believe it strikes the appropriate balance?
“What the Subcommittee has proposed is perfect. It introduces no new words of substance into the statute. All of the existing § 112(f) law developed since 1952 would be preserved. It amends the statute only be removing limitations… Seldom can the effects of a statutory change be summarized in a manner that is this clear and this concise.”
Are there any other potential changes you think we should consider pairing with our 101 fix, like an experimental use exception for example?
“(1) Research Use Exemption… In general, therefore, the new § 271(j) would create an infringement exception that encompasses any philosophical or scientific inquiries where the object is gaining knowledge regarding the patented invention… These exclusions are premised on the understanding that the public must be able to develop a complete understanding of the patented subject matter and be able to use that understanding to advance the useful arts…
“(2) Post Issuance Review Reforms…
“B. Permitted Scope for Review and Prior Art… 35 U.S.C. § 311(b) is amended to authorize an inter partes review on any ground that could be raised under as an invalidity defense in a patent infringement action so long as the petition for the inter partes review is filed within 9 months after the issue date of the patent…
“E. Limitation in Case of a Prior Adjudication… This new subsection prevents the United States Patent and Trademark Office from undertaking an inter partes review of a claim that has been the subject of a final written determination in a prior inter partes review proceeding or has been adjudicated not to be invalid in a prior civil litigation or International Trade Commission proceeding…
“(4) Subject Matter Eligibility Limited to Practically Useful Applications of Technology to an Area of Human Endeavor… For the purposes of amended § 101, areas of human endeavor are those where humankind has intervened in the natural world to produce applications of laws, phenomena, or ideas exhibiting, which applications must exhibit some specific and substantial utility to be eligible for patenting…”

Scott Partridge
Scott Partridge, Immediate Past Chair, Intellectual Property Law Section, American Bar Association
As [a] practitioner[] in the field, how has the current state of patent eligibility impacted the ability of your clients to receive patent protection for new, innovative and emerging technologies?
“The current jurisprudence on patent eligibility is confusing, creates uncertainty as to the availability and enforceability of patent assets, arguably risks the incentive to innovate provided by patents in technologies in which U.S. industry has historically led the world, and potentially places the U.S. in a less advantageous position on patent protection than our leading competitor nations…
“The effects of the current interpretation of section 101, however, are clear in one respect. Patents invalidated on the basis of eligibility in the U.S. are found to eligible in other countries. In a global economy, this puts at risk U.S. leadership in innovation.”
[I]f you were advising a client who wants to undertake hundreds of millions, if not billions, of dollars in research and development about what an “abstract idea” “law of nature” or “natural phenomena” is, what would you say? Could you give them any level of certainty or predictability?
“If hundreds of millions, if not billions, of dollars are in play in any given situation, it would be a rare circumstance to rely on patent eligibility as a basis for such a business decision…
“Outside of the US Patent and Trademark Office, it is reasonable to assume that Federal Circuit judges see more patent eligibility cases than any other lawyers or others involved in the intellectual property field, and they are asked to apply Supreme Court precedent to these cases. If they cannot decide these cases with any level of certainty or predictability, then certainly lawyers and clients are likely even less able to do so…
“Furthermore, the recent USPTO Guidelines on patent eligibility indicate the difficulty the US Patent and Trademark Office faces in attempting to implement current patent eligibility jurisprudence. Would these Guidelines even be necessary but for the lack of certainty and predictability in eligibility jurisprudence?… Would this be necessary if the current jurisprudence itself offered a clear and certain test that could be objectively applied across the board? The answer is no.”

Peter O’Neill
Peter O’Neill, Executive Director, Cleveland Clinic Innovations
What is the public policy value in encouraging investment, research, development and innovation in life sciences and precision medicine?
“The ability to tailor a treatment to a specific patient based on the patient’s genomic or other patient-specific features has profound implications to improve effectiveness and cost of how healthcare is delivered…
“Bringing a new product to market for patient care requires regulatory approval, clinical demonstration, production scale-up, sales growth, and countless other steps, all of which require significant investment. Intellectual property, including patent protection, is a critical part of that process and often is a key criterion which investors consider when deciding to make an investment in a new technology.”
Some have claimed we want to allow the patenting of human genes as they exist in the body. That’s false. However, I do think there’s value in promoting researchers and innovators to isolate human genes and apply that isolation to personalized treatment. Can you explain for us why that’s valuable?
“Personalized treatment (or targeted treatment) is the practice of detecting a genetic cause or driving factor for disease in an individual, and then using that information to identify a treatment for the disease that is specific to the genetic makeup…
“This practice allows the patient and their physicians to utilize treatments that have the highest likelihood of shrinking or stabilizing cancer tumors, as opposed to spending time trying treatments that the cancer tumor will not respond to…
“The targeted treatment options will continue to increase as we learn more about the genetic makeup of different cancer tumors and develop therapies specifically to treat these genetic variants.”
Looking forward ten to fifteen years, if we don’t correct the current state of patent eligibility what is the negative impact that American patients will experience?
“Under the status quo, there is significant uncertainty about what technologies are eligible for patents that can withstand legal challenges…
“That uncertainty makes it more difficult to bring new products to market, where they can be available for patient care. Particularly in the area of diagnostics in the life sciences, where Cleveland Clinic patents have been challenged and overturned in court, the uncertainty of patent protection makes it less likely we and other inventors will make the investments to make new advances commercially available.”
Image Source: Deposit Photos
Image ID: 2075803
Copyright: iqoncept

![[IPWatchdog Logo]](https://ipwatchdog.com/wp-content/themes/IPWatchdog%20-%202023/assets/images/temp/logo-small@2x.png)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Artificial-Intelligence-2024-REPLAY-sidebar-700x500-corrected.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/UnitedLex-May-2-2024-sidebar-700x500-1.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Patent-Litigation-Masters-2024-sidebar-700x500-1.jpg)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/WEBINAR-336-x-280-px.png)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/2021-Patent-Practice-on-Demand-recorded-Feb-2021-336-x-280.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/Ad-4-The-Invent-Patent-System™.png)






Join the Discussion
5 comments so far.
Stan Wellington
October 13, 2019 10:04 amThe people Tillis will help are the ones giving him money, a cave out for phama and Big Tech will happen and the ones that will be left out will be the small patent holders.
Tillis has received $262,126 in donations since 2014 (when he came into office), with the largest, $22,500, coming from Genentech. Other large donors include Biogen ($20,000), Pfizer ($20,000) and Amgen ($19,500). Tillis serves on the Committee on Agriculture, Nutrition and Forestry, Committee on Armed Services, Committee on the Judiciary, Committee on Veterans’ Affairs, and the Special Committee on Aging.
It has been noted that Tillis is something of a defender of the biopharma industry. Early on, in 2015, he worked hard to compromise on a patent reform bill by carving out an exemption related to an FDA review process.
The KHN database is searchable. For example, Sen. Mitch McConnell (R-Ky), has received $972,999 since 2007 from biopharma. The largest single donor is Amgen, which donated $81,000. Sen. Chuck Grassley (R-IA) has received $296,000, with Amgen also the largest donor ($56,000).
The New America only helps Big Business. New inventors will need to look to other countries for patent protection. The American Government has turned their backs on patent holders as they collect their checks from Big Tech and Big Pharma. The new Corporatocracy will be the death of Capitalism.
CHINA WINS!!!!
August 28, 2019 09:33 pm@ b
You know nothing. The devil is in the details. Big Tech pays big just like Big Pharma. Big fish eats small fish, IPs are done.
B
August 26, 2019 12:34 pm“Puppets like Sen. Tilis will do as they have paid him to do . . . . ”
FYI, I’ve talked to Senator Tillis, and I know what the latest proposal to s101 is.
Everyone except the efficient infringement people loved the 101 revisions.
The 112(f) revision is Williamson v. Citrix in statute form, and I personally have no problem with it as the previous “presumptions” created were murky. ALWAYS provide a thorough 112(a) disclosure even assuming your claims will never be interpreted under 112(f).
To me the only problem is the proposed 100(k), and only the word “technological”
China WINS!!!
August 23, 2019 08:57 amFAANG runs the rules. Puppets like Sen. Tilis will do as they have paid him to do and that is to keep patents weak so Silicon Valley Elites can continue to steal patents from patent holders with out having to pay licensing fees. NO point filing for a patent protection in the United States its game over.
Josh Malone
August 22, 2019 10:44 amBob Armitage says that the written description requirement of 112(a) worked to invalidate the claim if “stunning breadth”.
If 112(a) works then why does he persist in monkeying with 112(f)?
If in his mind the USPTO cannot follow 112(a) why does he think they will follow his new scheme?
I am shocked that this nonsense is getting traction. Inventors have already largely abandoned the U.S. patent system since the AIA.
Eliminating use of functional terms from our claims would make it impossible to obtain rights to the full scope of our inventions. It would leave holes of “potentially stunning breadth” in our patents.
This “perfect” proposal would do more damage than he caused with his contributions to the AIA.