“Moderna’s COVID-19 pledge positions it well to grow the new mRNA market its business is founded upon, capitalize on licensing deals, and restore all enforcement rights “post pandemic.”
 As Pfizer and BioNTech announced this week that results of their COVID-19 vaccine have exceeded expectations, the technology behind the vaccine (messenger RNA, or mRNA) has taken center stage. Last month, Moderna Therapeutics, one of the global leaders in the race to produce a COVID-19 vaccine using mRNA, made the following statement regarding enforcement of its patents:
As Pfizer and BioNTech announced this week that results of their COVID-19 vaccine have exceeded expectations, the technology behind the vaccine (messenger RNA, or mRNA) has taken center stage. Last month, Moderna Therapeutics, one of the global leaders in the race to produce a COVID-19 vaccine using mRNA, made the following statement regarding enforcement of its patents:
We feel a special obligation under the current circumstances to use our resources to bring this pandemic to an end as quickly as possible. Accordingly, while the pandemic continues, Moderna will not enforce our COVID-19 related patents against those making vaccines intended to combat the pandemic. Further, to eliminate any perceived IP barriers to vaccine development during the pandemic period, upon request we are also willing to license our intellectual property for COVID-19 vaccines to others for the post pandemic period.
This post examines why Moderna made this patent pledge by examining its mRNA technology, go-to-market status, patent landscape, and market position.
Moderna’s Technology
Moderna (appropriately listed in NASDAQ as “MRNA”) was founded by Flagship Pioneering in 2010 and utilizes mRNA molecules to cause a recipient’s own cells to express targeted proteins for a desired therapeutic effect. Moderna claims that such therapies can be used to treat a wide range of conditions beyond infectious diseases such as COVID-19.
How does it work for COVID-19?
Moderna’s COVID-19 vaccine introduces “mRNA-1273” molecules into the recipient via a delivery system that protects the mRNA in the bloodstream and allows for entry into cells. Once inside the cells, the mRNA-1273 molecules act like computer code and instruct the cells to produce the characteristic COVID-19 spike proteins. The immune system identifies the spike proteins as antigens and builds its defenses against those antigens in preparation for the potential exposure to the actual COVID-19 virus.
This is “a fundamentally different approach” than traditional vaccines that inject entire viruses, either alive (attenuated) or dead, into the recipient to trigger the desired immune response. Moderna claims its mRNA vaccine technology offers potential advantages over traditional vaccines in terms of “efficacy, speed of development, and production scalability and reliability . . ..”
Moderna believes its mRNA technology could become the foundation for a new therapeutic modality that transforms the drug development and biologics space for decades to come, similar to the disruption that Biogen, Amgen and Genentech made in the ’80s when they developed biologic protein therapies.
Moderna’s Go-To-Market Status
Moderna was focused on mRNA-based vaccine development and had already built a growing mRNA-based pipeline for various indications including infectious diseases, cancer, cardiovascular diseases and rare genetic diseases well before the pandemic arrived.
Fig. 1: Moderna is poised to flood the COVID-19 vaccine market, having previously developed several other mRNA-based therapies for other indications.
It was therefore well-positioned to produce an mRNA-based vaccine candidate (mRNA-1273) and begin the regulatory approval process at the outset of the pandemic. Indeed, Moderna was the first in the world to do so within about two months from the date COVID-19’s molecular blueprint was known, and is now widely viewed as one of the top contenders in the race to produce a viable COVID-19 vaccine.
As of the date of this writing, the U.S. Government has agreed to purchase 100 million doses of Moderna’s COVID-19 vaccine, with the opportunity to obtain an additional 400 million doses. Moderna has also struck a deal with the European Commission to supply an initial 80 million doses to Europe with an option to purchase 80 million more, as well as with Japan to supply 50 million doses, and with Qatar as well.
Moderna has expressed confidence in its ability to manufacture vaccine in large quantities to meet demand, including 20 million doses of mRNA-1273 by the end of this year, and between 500 million and 1 billion doses in 2021. Reports also indicate that Moderna has received $1.1 billion in deposits for supplies of the vaccine.
Accordingly, should its vaccine be approved as safe and effective, Moderna appears poised to flood the COVID-19 vaccine market, recoup a hefty payday at launch, and be positioned to supply its vaccine under existing and new contracts throughout the world on a going-forward basis.
Patent Landscape for Moderna/mRNA
Moderna and its investors have spent billions of dollars on mRNA research and Moderna has sought and obtained hundreds of patents in the United States, Europe, Japan, and other jurisdictions to protect that technology, with several hundred patent applications pending. Moderna has identified at least seven U.S. patents that it alleges protect the subject mRNA-1273 technology, and has numerous patent applications pending, including dozens in the United States alone.
Some third-party patents appear to pose challenges for Moderna. For example, the biotech has been in an ongoing patent dispute with Arbutus Biopharma, which owns certain patents relating to lipid-nanoparticle “delivery systems” for mRNA molecules. These delivery systems essentially wrap the molecules in balls of fat to both disguise them, thereby preventing attack by the immune system, and to allow the target cells to readily ingest the mRNA molecules.
Moderna had a limited sublicense to those disputed patents and (before the pandemic) sought to invalidate the patents through three USPTO inter partes review proceedings that have had mixed results (one win that was reversed on appeal and is on remand, one partial win that is on appeal, and one loss that is on appeal).
There have also been reports that the National Institutes of Health (NIH) has an ownership interest in Moderna’s mRNA-1273-related patents and has filed its own patents relating to the technology, raising potential ownership, infringement, and validity issues.
There are also reports that Moderna failed to report Department of Defense funding (through DARPA) underlying many of its patents, which has potentially negative implications including under the Bayh-Dole Act that provides, among other things, the U.S. Government with certain rights to use inventions it helps fund.
Moderna has also notified its investors of the existence of “many issued and pending third-party patents” that it may need to license or that may be asserted against Moderna.
Despite these issues, Moderna has nonetheless expressed confidence that it is “not aware of any significant intellectual property impediments for any products we intend to commercialize, including mRNA-1273.”
Potential Economic Rationales
Moderna and several other teams are developing mRNA-based-vaccines to combat COVID-19 including Pfizer (with BioNTech), Arcturus (with Duke Medical School), Sanofi (with Translate Bio), Abogen (with Walvax Biotechnology), Chulalonkorn University, CureVac, and others.
Other teams are developing traditional, non-mRNA-based vaccines, such as “viral vector” and other vaccines,including Johnson & Johnson, AstraZeneca, Merck, and others, and yet other teams are developing protein-based and other vaccines that also do not utilize mRNA technology. A comprehensive list of developers and the status of their programs can be found here.
The COVID-19 vaccine market (“Covid Market”) is therefore comprised of numerous companies utilizing different technologies, several of which (importantly) utilize the new and non-traditional mRNA technology.
Fig. 2: Moderna is expected to be a leading entrant into the COVID-19 vaccine market, comprised of other drug makers utilizing mRNA, traditional, non-mRNA-protein, and other vaccine technologies.
Recall that in the bigger picture, Moderna wants mRNA-based therapies to fundamentally transform the drug development space for decades to come. The pandemic—a black swan event—appears to have opened that door. Here’s why:
As of the date of this writing it appears the first two market entrants into the Covid Market may be Pfizer (reporting a 90% vaccine effectiveness rate from its trials) and Moderna (with its interim trial results expected shortly), each vaccine utilizing mRNA technology.
No matter who follows after that, should the first two COVID-19 vaccines be mRNA-based, this development would hammer a very large stake into the ground, marking mRNA-technology as a viable alternative in the drug development space.
From an execution standpoint, success of the mRNA COVID vaccines could pave the way for future therapies in the infectious disease space and potentially other mRNA therapies, including the therapeutic candidates that Moderna has steadily developed since 2010. With its current pipeline and patent portfolios, Moderna appears to be very well positioned to capitalize on such downstream markets.
Moderna’s pledge not to enforce its patents against companies that use mRNA to make COVID-19 vaccines serves that end. The hands-off approach allows other market participants to aggressively pursue mRNA-based therapies for COVID-19 without fear of suit. This increases the chances of a viable mRNA vaccine getting to market. It also may result in developers investing further in the technology to develop other mRNA therapies post-pandemic for other indications.
But why would Moderna want to encourage more market participants and potential competitors? In a fledgling market, it’s pure economics.
More market participants will help drive the diffusion of the new mRNA technology into the drug development ecosystem and help build infrastructure for mRNA-based technologies within that new ecosystem.
This rationale is supported by a number of sources, including a 2019 Cambridge University study that found tech diffusion and infrastructure building to be the “primary” drivers behind companies’ decisions to make patent pledges, including by companies such as Tesla Motors, which made such pledges to spur long-term growth in new markets.
Fig. 3: Moderna may hope to encourage participation by other players in the new mRNA market to drive mRNA tech diffusion, infrastructure building and overall growth of the new tech platform.
Recall also that Moderna is not giving much up under the terms of its self-prescribed pledge in that the pledge not to sue only applies to mRNA-based vaccine makers during the pandemic and that licensing is very much on the table pre and post pandemic.
With enforcement allowed post pandemic (a period of time ostensibly defined by Moderna), threat of suit will be very useful to leverage in such downstream licensing deals without a public relations backlash. The pledge is also limited in that it would not preclude Moderna from enforcing its patents for indications beyond COVID-19 at any time.
Accordingly, Moderna’s pledge positions it well to grow the new mRNA market its business is founded upon, capitalize on licensing deals, and restore all enforcement rights “post pandemic.”
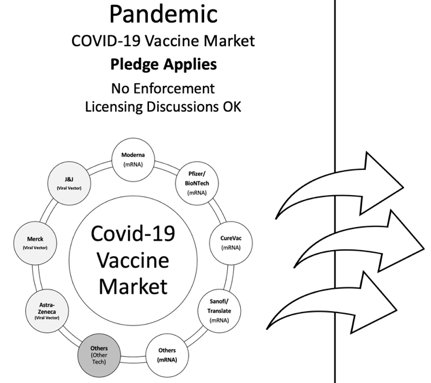
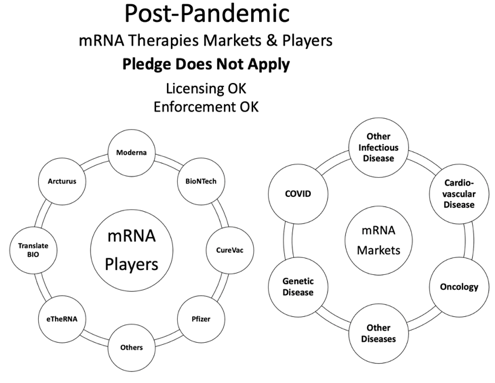
Fig. 4: The COVID-19 pandemic may have opened the door to fundamentally transforming the drug development space, and Moderna’s patent pledge may serve to cultivate the new mRNA market to yield longer-term gains.
Furthermore, Moderna’s promise not to enforce its COVID-19 related patents during the pandemic against COVID vaccine developers also sets a disarming tone in the space that may serve to deter others in the space from acting too defensively or aggressively.
For example, the pledge may serve to deter third party patent-holders from enforcing their own patents against Moderna in favor of licensing deals, for nothing more than fear of the potential public relations backlash. A cooperative posture by Moderna may also make the federal government less apt to assert ownership claims over certain of the mRNA patents it may have an ownership interest in, as well as obviate the need for compulsory licensing.
Moreover, aside from its obvious benefit to the fight against COVID-19, Moderna’s patent pledge also makes for good public relations. Their patent pledge statement, while technically narrow, was framed in altruistic terms in the context of the pandemic, and Moderna received significant praise from several industry groups including those calling for other companies to make similar pledges.
From another viewpoint, nothing good could come from Moderna enforcing its COVID-19 patents during the pandemic. Being an aggressor in this market, during the pandemic, risks incalculable damaging backfire, involving suits and countersuits by other patent-holders, USPTO inter partes invalidity proceedings, suits, and ownership claims by the federal government, compulsory licensing orders, all serving to potentially snuff out the fledgling market before it even has a chance to grow. Furthermore, should any delay in delivering the vaccine or other negative consequences flow from patent enforcement activity, the risk of public rancor could be catastrophic to the company’s reputation and its bottom line.
Many of the above rationales are supported by the comments that Moderna’s President Stephen Hogue made to the press regarding the pledge: “[W]e are quite studiously not asserting infringement. We’re doing the opposite of creating that kind of anxiety for folks. We’re not interested in using that IP to decrease the number of vaccines available in a pandemic.”
Indeed, this appears to be a shrewd policy decision that may serve to get the COVID-19 vaccine to market at warp speed whether by Moderna, Pfizer or others thereby helping the world, and fulfill Moderna’s broader mission by potentially establishing a transformational drug therapy platform that will advance healthcare and yield gains for the company for years to come.
Image Source: Deposit Photos
Author: Esbenklinker
Image ID: 75025297
*This article was updated on April 9, 2021

![[IPWatchdog Logo]](https://ipwatchdog.com/wp-content/themes/IPWatchdog%20-%202023/assets/images/temp/logo-small@2x.png)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/UnitedLex-May-2-2024-sidebar-700x500-1.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Artificial-Intelligence-2024-REPLAY-sidebar-700x500-corrected.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Patent-Litigation-Masters-2024-sidebar-700x500-1.jpg)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/WEBINAR-336-x-280-px.png)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/2021-Patent-Practice-on-Demand-recorded-Feb-2021-336-x-280.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/Ad-4-The-Invent-Patent-System™.png)






Join the Discussion
3 comments so far.
Dphar
December 24, 2020 11:58 amWhy would I want a vaccine that modifies the cells of my body? What else does it do? Yeah. I have trust issues.
dine
December 2, 2020 01:16 pm1. Moderna can pledge to supply free of cost vaccine as a charity but not patent charity as themselves are indulged in that infringement and to misguide the best they can do is to invite others to open multiple lawsuit to protect them for this infringement .
Jonathan Stroud
November 12, 2020 08:56 amThanks for this, Daniel, very insightful. I think Moderna’s pledge is very close to the Open COVID Pledge, the voluntary licensing pledge that would free up researchers’ and companies’ ability to race to a cure during the pandemic free of fear of liability. As you note, it’s pretty surgical in the time-limited nature of it and ultimately helps them AND the whole industry by increasing the ability to collaborate and build off of others’ advances in the short term, while not preventing securing those rights for the long term to any eventual product. I think it’s such a good idea that most of the companies have agreed to something similar, albeit under different names.