“It took 155 years for the USPTO to issue its first 5 million patents in 1991; it has taken less than 1/5 of that time to issue the next 6 million. Have we really become more inventive in the last 30 years or have we just got better at inventing patents?” – Tamir Amin, Initiative for Medicines, Access, and Knowledge

Rep. Carolyn Maloney
Yesterday, Representatives Carolyn Maloney (D-NY), Jerrold Nadler (D-NY) and David Cicilline (D-RI) asked Federal Trade Commission (FTC) Acting Chair Rebecca Kelly Slaughter to open a formal inquiry into pharmaceutical company AbbVie’s practices, which the representatives said have worked “to delay U.S. biosimilar entry for [AbbVie’s] blockbuster drug Humira.” The request was prompted by documents uncovered as part of an investigation being conducted by the House Committee on Oversight and Reform into the company. “Based on our review, these documents indicate that AbbVie delayed biosimilar competition for far longer than warranted by its own internal evaluations of the strength of its patent portfolio, which anticipated biosimilar entry no later than 2017,” said the letter.
Yesterday was also the House Committee on Oversight and Reform’s third hearing on “Unsustainable Drug Prices” featuring testimony from AbbVie CEO Richard Gonzalez. Maloney opened that hearing with claims that “drug companies are actively targeting the United States for price increases while lowering them in the rest of the world,” and said that the justifications the pharmaceutical industry offers for why they need to raise prices “simply do not hold water.” She also said that, while she thanked Gonzalez for being there, “it took more than a year and the threat of a subpoena” before the company voluntarily agreed to attend.
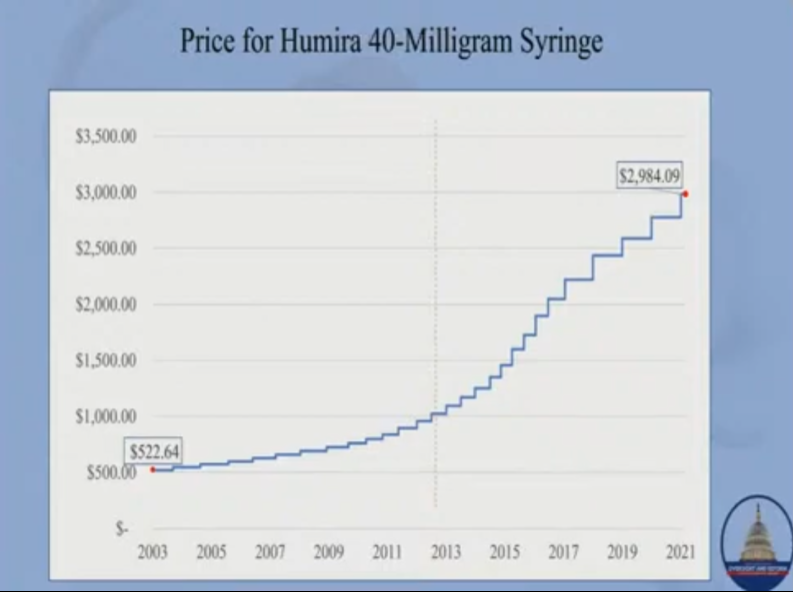 According to research conducted by the Committee, AbbVie has repeatedly raised the prices of its blockbuster drug Humira, which is used to treat rheumatoid arthritis, and Imbruvica, used to treat cancer. It presently costs $77,000 a year for Humira, which is 400% more than when the drug was launched. The price of a syringe of Humira started at $500, now it’s $2,984. AbbVie and Janssen charged even more for Imbruvica—over $181,000 for a year’s supply, which is 74% more than when the drug was launched in 2013. “These prices are outrageous and unfair,” Maloney said. “More outrageous is that Americans are the only ones paying them.”
According to research conducted by the Committee, AbbVie has repeatedly raised the prices of its blockbuster drug Humira, which is used to treat rheumatoid arthritis, and Imbruvica, used to treat cancer. It presently costs $77,000 a year for Humira, which is 400% more than when the drug was launched. The price of a syringe of Humira started at $500, now it’s $2,984. AbbVie and Janssen charged even more for Imbruvica—over $181,000 for a year’s supply, which is 74% more than when the drug was launched in 2013. “These prices are outrageous and unfair,” Maloney said. “More outrageous is that Americans are the only ones paying them.”
Humira is $1,000 higher in the United States than in Canada, Japan Korea and the UK. AbbVie has been dropping prices elsewhere while raising them in the United States.
Maloney said the Committee’s investigation also uncovered evidence that AbbVie has “exploited the U.S. patent system and engaged in anticompetitive practices to extend its monopoly pricing.” This was the impetus for the letter to the FTC.
She added that AbbVie makes “essential, lifesaving products” and “we want drug companies to be successful, but abusive, unfair pricing and anticompetitive practices mean these medicines are out of reach for too many Americans.”

Richard Gonzalez
In his testimony, Gonzalez called for “industry, government and healthcare plans” to “come together to significantly reduce out of pocket cost for patients.” He said that “Medicare Part D patients are bearing too much of the out-of-pocket cost and there is no cap on drug spending.” However, he acknowledged that AbbVie does consistently increase prices in the United States while decreasing price in other countries because “that is how it works.”
Republican congress members criticized Democrats for dismissing the undeniable success of Operation Warp Speed and the U.S patent system and vilifying the drug companies that helped to get the country on the road to recovery. “Cases and fatalities have plummeted, we’re on the road to recovery, and the catalyst has been intellectual property. Our patent system allows for robust innovation,” said Committee Ranking Member James Comer (R-KY).
Representative Virginia Foxx (R-NC) pointed to the example of “price controls” threatened by Senator Bernie Sanders (D-VT) on a collaboration between Sanofi and the U.S. government to develop a vaccine for Zika virus as evidence of what such measures lead to. Sanofi ultimately said it would not seek a license to develop the vaccine, and there is still no vaccine for Zika. Foxx wondered, if the threat of price controls on one potential product chilled innovation so starkly, what would happen if such measures were extended?
Craig Garthwaite, the Herman Smith Research Professor in Hospital and Health Services, Kellogg School of Management at Northwestern University, said in his statement that the U.S. government should focus on improving the health insurance market to reduce pharmaceutical prices:
[O]nerous cost sharing in many existing insurance contracts can unwind the value of the insurance product – something that could be particularly concerning if patients are not fully aware of the terms of the contract when buying insurance. Some of this onerous cost sharing results from the growing size of rebates in the market while others are a feature of the insurance contract – in particular those in the Medicare Part D program. Policymakers should work to address these issues.
Garthwaite also said that government should play a bigger role in fostering competition in the pharmaceutical space and ensuring that periods of market exclusivity are “time limited and followed by robust entry and competition from new firms.”
Tahir Amin, Co-Founder and Co-Executive Director, Initiative for Medicines, Access, and Knowledge, however, said that the United States has not just a drug pricing crisis, but a drug patenting problem:
Last week, the USPTO issued its 11 millionth patent. It took 155 years for the USPTO to issue its first 5 million patents in 1991; it has taken less than 1/5 of that time to issue the next 6 million. Have we really become more inventive in the last 30 years or have we just got better at inventing patents because our patent system no longer is stringent enough?

Tahir Amin
Amin said that his organization’s research shows that 1,310 patent applications have been filed on the top 10 selling drugs in the United States and 692 patents granted total. That amounts to 131 patent applications and nearly 70 granted patents per drug, and provides an average monopoly protection of 38 years. Abbvie has filed 422 patent apps on Humira and Imbruvica alone, 90% of which were filed after the drug was first approved in 2002. The company has 130 granted patents for Humira and 39 years of protection. “Patent settlement agreements have allowed AbbVie to keep competitors out of the market until 2023,” Amin said.
Amin also said that roughly 34 million American adults know one friend or family member who has died in the past five years because they could not afford treatment, and that number is double for people of color.
Yesterday’s 106-page letter to the FTC includes a number of exhibits and documents that the authors claim demonstrate “that AbbVie’s own evaluation of the strength of its patent portfolio projected that biosimilar entry would occur much earlier than the 2023 biosimilar entry date agreed to by AbbVie and its competitors.” They further urged the FTC to:
[S]crutinize AbbVie’s agreements and negotiations with biosimilar competitors for their compliance with U.S. law, including whether transfers of value occurred outside the express terms of each written agreement. In addition, we request that the FTC examine whether AbbVie engaged in other anticompetitive conduct to maintain its market share and pricing power for Humira, such as shifting patients to a higher concentration formulation of the drug and using bundled rebates to exclude rivals of preferred formulary positions.

![[IPWatchdog Logo]](https://ipwatchdog.com/wp-content/themes/IPWatchdog%20-%202023/assets/images/temp/logo-small@2x.png)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/UnitedLex-May-2-2024-sidebar-700x500-1.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Artificial-Intelligence-2024-REPLAY-sidebar-700x500-corrected.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Patent-Litigation-Masters-2024-sidebar-700x500-1.jpg)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/WEBINAR-336-x-280-px.png)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/2021-Patent-Practice-on-Demand-recorded-Feb-2021-336-x-280.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/Ad-4-The-Invent-Patent-System™.png)






Join the Discussion
7 comments so far.
Anon
May 21, 2021 12:43 pmOne aspect only hinted at in the article (and for which I have previously proposed) is a full “open kimono” of the entire Pharma world.
Let that entire mechanism be out into the full sunlight of public scrutiny.
Having assisted discovery in a Pharma litigation matter long ago, I can guarantee you that the shadows (and skullduggery therein) are DEEP.
Anon
May 21, 2021 12:22 pmAnd yet another example of Big Pharma in the sights of those seeking dismantling of the strengths of Big Pharma.
While indeed different than the COVID land grab (and in fact on a topic that I have also called out Big Pharma for in the past), those who may want to strike the Ostrich pose of “Pharma is too powerful to let the Biden administration apply the full force of Woke Liberal Left ‘Equity,’ may want to recalibrate.
Model 101
May 21, 2021 12:06 pmI’m quoting Robert Armitage from the 2019 Senate Hearings on Patent Eligibility.
Pro Say @ 4 Well said indeed!
I say again only louder… LEAVE THEM ALONE! (please!)
Pro Say
May 21, 2021 09:27 amModel 101: “The cost of a drug you can’t get Is infinite.”
. . . with the cost of a life-saving drug you can’t get being death.
George
May 20, 2021 07:33 pm@Model 101
Was it Jonas Salk that said that????
George
May 20, 2021 07:32 pm“Have we really become more inventive in the last 30 years or have we just got better at inventing patents?””
LOL!
Model 101
May 20, 2021 12:24 pmThe cost of a drug you can’t get Is infinite.
Leave them alone..