“In total, the data show a decline in ANDA case filings of about 36% from 2017 to 2021. The same trend is noticed with the number of the patents involved in these cases.”
Last week, Patexia released its second annual ANDA Litigation Intelligence Report in which we covered the rankings, statistics and comprehensive analysis of abbreviated new drug application (ANDA) and Hatch-Waxman stakeholders. We couldn’t help but notice the decline in ANDA filing activity that has begun, after reaching its peak in 2018. Year-over-year comparison of the ANDA data, as seen in the following chart, shows the trend in the last four years. We observe a considerable decline from 2018 to 2019 (a decline of 4.2 cases/month) followed by a smaller decline from 2019 to 2020 (2.7 cases/month) and the sharpest decline from 2020 to 2021 (5.3 cases/month) indicated by the data analyzed for the first half of 2021. In total, this represents a decline in ANDA case filings of about 36% from 2017 to 2021. The same trend is noticed with the number of the patents involved in these cases.
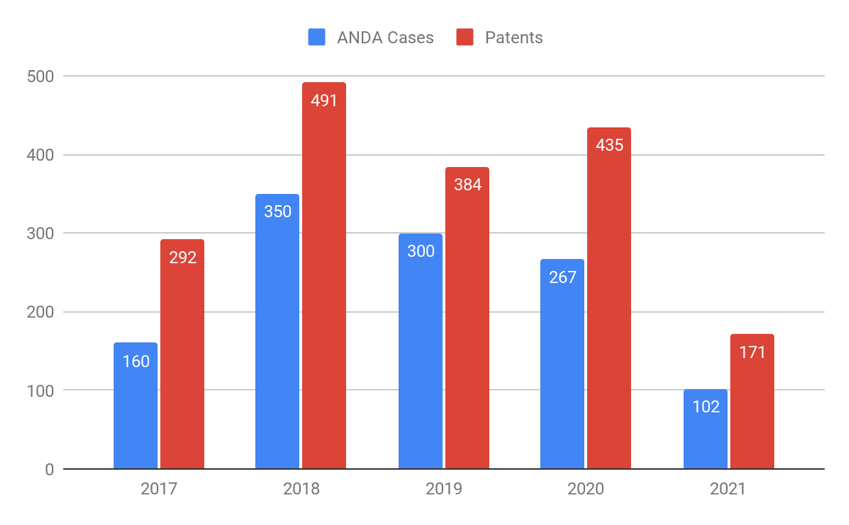 The accuracy of the data has been improved this year by crawling and parsing more legal documents related to Hatch-Waxman cases, which resulted in identifying 1,471 attorneys who were involved in a total of 1,179 ANDA cases, filed in a four-year period from July 1, 2017 through June 30, 2021. We have also evaluated all stakeholders in Hatch-Waxman cases, including ANDA attorneys, local counsels, law firms, pharmaceutical companies and judges, ranking them based on their activity and performance. While ANDA cases make up a small fraction of all patent cases (sometimes less than 10% of the annual district court patent litigation cases), they prove to be of a great importance for law firms and companies as well, because of high potential earnings that come from brand and generic drugs, so it’s worth examining the possible reasons for the decline.
The accuracy of the data has been improved this year by crawling and parsing more legal documents related to Hatch-Waxman cases, which resulted in identifying 1,471 attorneys who were involved in a total of 1,179 ANDA cases, filed in a four-year period from July 1, 2017 through June 30, 2021. We have also evaluated all stakeholders in Hatch-Waxman cases, including ANDA attorneys, local counsels, law firms, pharmaceutical companies and judges, ranking them based on their activity and performance. While ANDA cases make up a small fraction of all patent cases (sometimes less than 10% of the annual district court patent litigation cases), they prove to be of a great importance for law firms and companies as well, because of high potential earnings that come from brand and generic drugs, so it’s worth examining the possible reasons for the decline.
The trend of decline in ANDA is different from the larger trend observed in recent years (i.e., increased activity in district court patent litigation, International Trade Commission [ITC] Section 337 and inter-partes review [IPR]). Here, we should underline that ANDA cases are unique among patent litigation matters because the act of infringement itself is the submission of an application to the Food and Drug Administration (FDA) to market a generic drug in the future, rather than the “classical” infringement case, which involves making, using, offering to sell, selling, or importing a patented product.
A pharmaceutical company that wants to develop a generic version of the previously patented drug should file an ANDA not only to demonstrate that the new drug is chemically or pharmaceutically equivalent, but also to prove the “bioequivalence”, meaning that the generic drug will work in the body in the same way and be as safe and effective as the brand drug. From this information, we deduce that the reasons for the decline in ANDA litigation should be explored first of all in the patent activity, more specifically the drug patents granted 20 years ago. This number might be lower for that period, so today we have less patented drugs that are expiring.
Furthermore, we know that one branded drug does not have an equal profitability to another, which results in accumulated interest in specific drugs and multiple ANDA cases filed over them. Here, we can mention as an example Abilify, which is a top-selling antipsychotic prescription in the United States. The company that manufactures the drug, Otsuka, earned revenue of $4.9 billion in 2014 alone. Another example would be Oxycontin, used for the treatment of moderate to severe pain. This drug has generated approximately $30 billion in revenue for its manufacturer Purdue Pharma over the years. Both of these drugs have been litigated in more than 60 and 50 ANDA cases, respectively, in the past years. This makes us want to look into the data of the drug patents that have expired recently to see if we can find something similar in profits that might gather the attention of the top generic manufacturer pharmaceutical companies.
In a list provided by Pharmatutor we don’t see drugs with record sales like the ones mentioned above that could be litigated in multiple cases by different companies. The only exception is Truvada from Gilead Sciences, which is used as a treatment for HIV. During 2019 alone in the United States, they made $2.64 billion in sales. The patent for emtricitabine, one of the two main components of Truvada, expires in September of 2021, so it will be very interesting to see how this will reflect in the number of ANDA cases for 2021 in total.
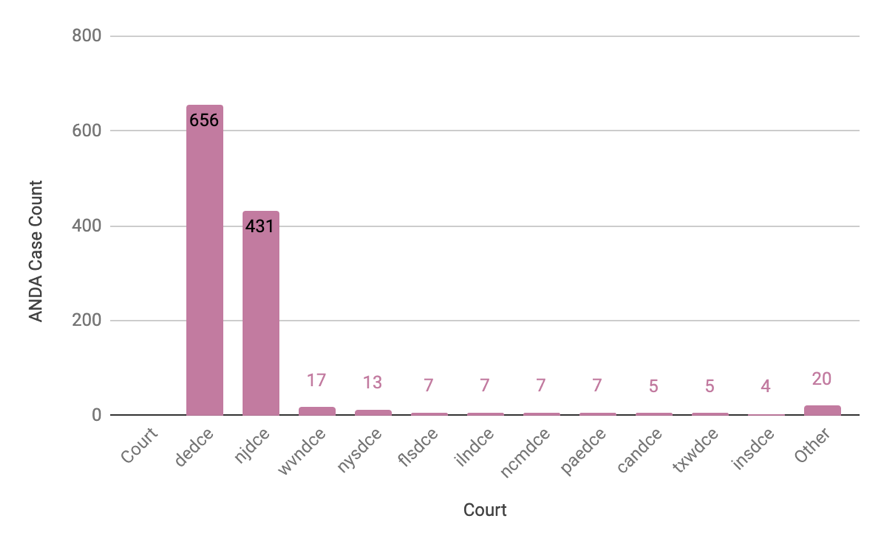
ANDA data from our ANDA Litigation Intelligence 2021 Report show even more interesting statistics, such as the distribution of case filings by jurisdictions. The majority of generics companies in the United States are based or incorporated in Delaware or New Jersey, so it doesn’t come as a surprise that New Jersey and Delaware account for 92% (1,087/1,179) of cases filed during the period of our study. The rest is scattered throughout a variety of district courts from 16 other states (NY, WV, FL, NC, PA, IL, IN, TX, CA, CO, LA, NV, CT, MI, UT, and VA) as seen in the chart below:
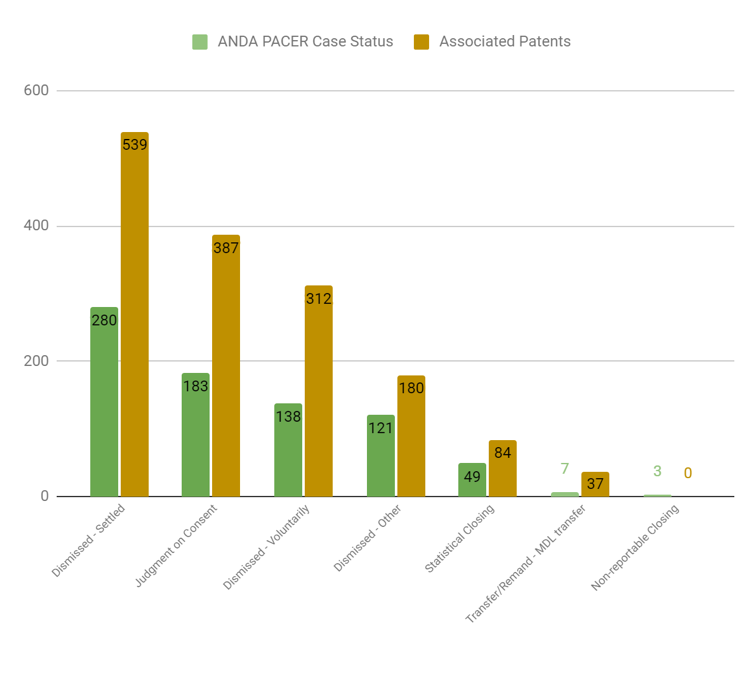
 Our deeper analysis of the legal documents related to Hatch-Waxman cases reveals more interesting observations, especially after we evaluated the outcomes of these cases. It is worth mentioning that ANDA cases are less likely to end in a settlement compared to other types of patent litigation. It is notable that the majority of ANDA cases are dismissed for one reason or another; a smaller portion go to a judgment of some kind. The review of these judgment decisions suggests that the claimant in an ANDA case has more chance to win than in other patent litigation cases. Below is a chart that shows the PACER status of ANDA Cases filed and decided between July 2017 through June 2021:
Our deeper analysis of the legal documents related to Hatch-Waxman cases reveals more interesting observations, especially after we evaluated the outcomes of these cases. It is worth mentioning that ANDA cases are less likely to end in a settlement compared to other types of patent litigation. It is notable that the majority of ANDA cases are dismissed for one reason or another; a smaller portion go to a judgment of some kind. The review of these judgment decisions suggests that the claimant in an ANDA case has more chance to win than in other patent litigation cases. Below is a chart that shows the PACER status of ANDA Cases filed and decided between July 2017 through June 2021:
 The outcome of these cases and how they favored either the plaintiffs or the defendants was essential in determining the activity and performance of 364 companies, 1,471 attorneys and 243 law firms, out of which we have also identified the ones that acted as local counsel, especially in New Jersey and Delaware, where some attorneys and law firms were very active during the period our study covered.
The outcome of these cases and how they favored either the plaintiffs or the defendants was essential in determining the activity and performance of 364 companies, 1,471 attorneys and 243 law firms, out of which we have also identified the ones that acted as local counsel, especially in New Jersey and Delaware, where some attorneys and law firms were very active during the period our study covered.

![[IPWatchdog Logo]](https://ipwatchdog.com/wp-content/themes/IPWatchdog%20-%202023/assets/images/temp/logo-small@2x.png)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Patent-Litigation-Masters-2024-sidebar-early-bird-ends-Apr-21-last-chance-700x500-1.jpg)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/WEBINAR-336-x-280-px.png)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/2021-Patent-Practice-on-Demand-recorded-Feb-2021-336-x-280.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/Ad-4-The-Invent-Patent-System™.png)






Join the Discussion
No comments yet.